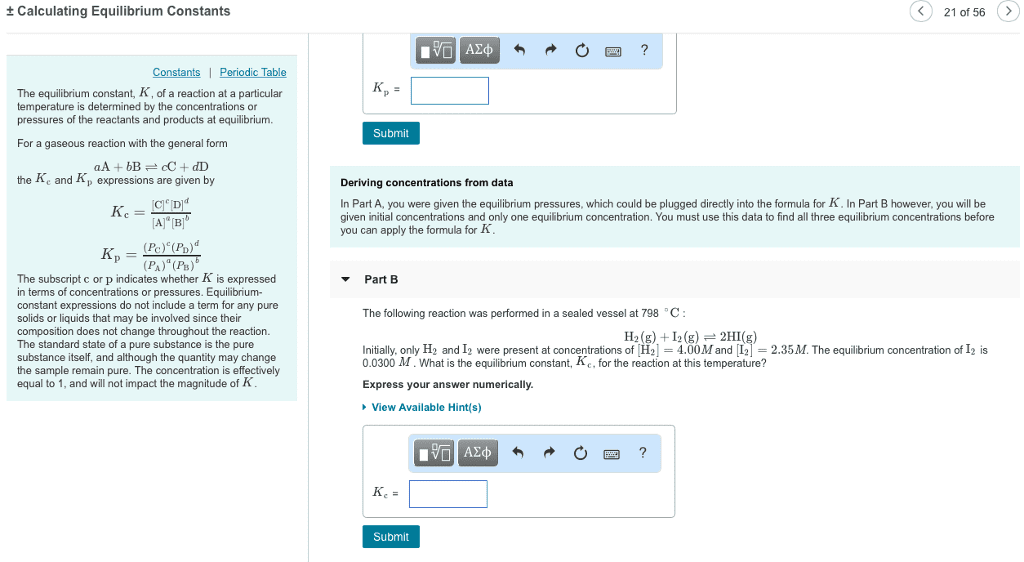
1) 2)
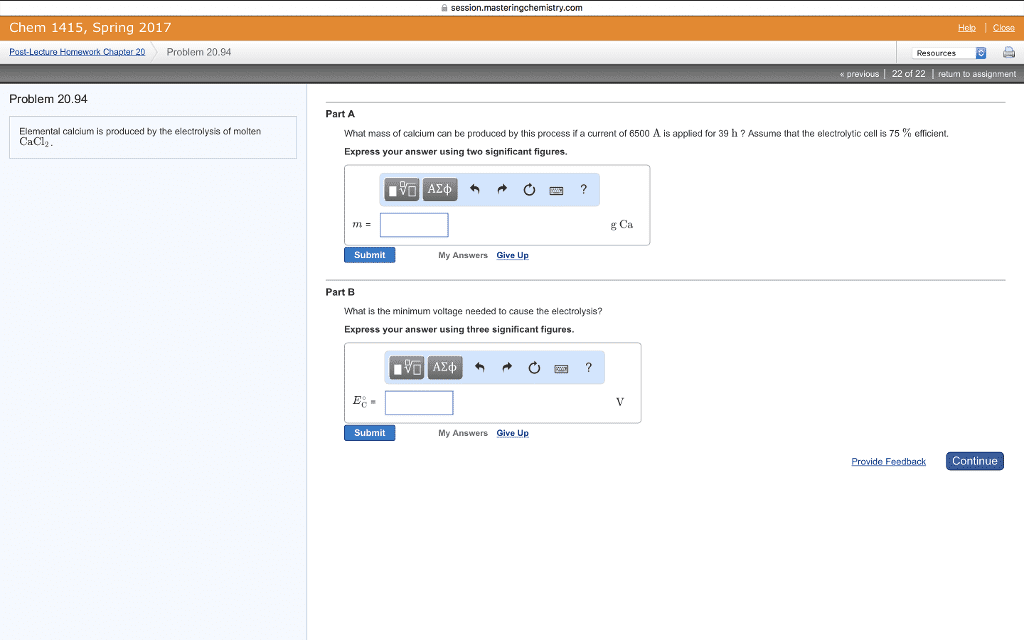
2)
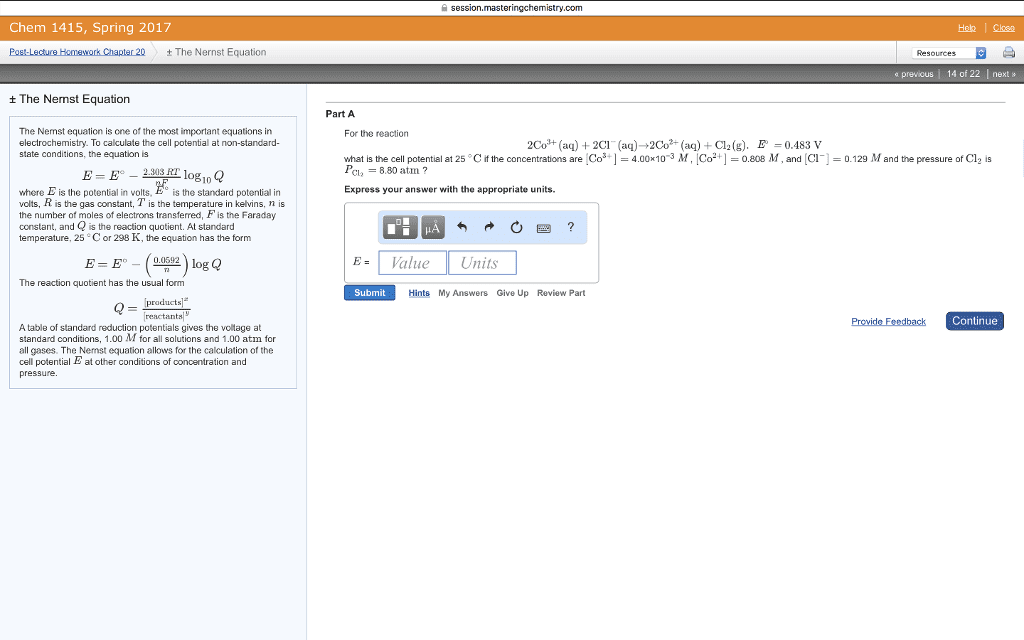
Chem 1415, Spring 2017 PostLecture Homework Chapter 20 t The Nernst Equation t The Nemst Equation The Nemst equation is one of the most important equations in electrochemistry. To calculate the cell potential at non-standard- state conditions, the equation is 2.303 RT og10 Q where Eis the potential in volts, E is the standard potential in volts, R is the gas constant, T is the temperature in kelvins, n is the number of moles of electrons transferred, Fis the Faraday constant, and Q is the reaction quotient. At standard temperature, 25 C or 298 K, the equation has the form log Q The reaction quotient has the usual form products A table of standard reduction potentials gives the voltage at standard conditions, 1.00 M for all solutions and 1.00 atm for all gases. The Nernst equation allows for the calculation of the cell potential E at other conditions of concentration and pressure. ngchemistry.com Resources previous 14 of 22 next Part A For the reaction 2Co H q) 2Cl (aq)- 2Co (aq) Cl2(g). E 0.483 V what is the cell potential at 25 Cif the concentrations are Cos 4.00x10 3 M, ICo +1 0.808 M and C 0.129 M and the pressure o Cl2 is Pola 8.80 atm Express your answer with the appropriate units. E Value Units Submit Hints My Answers Give Up Review Part Continue