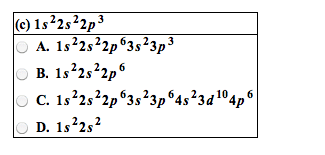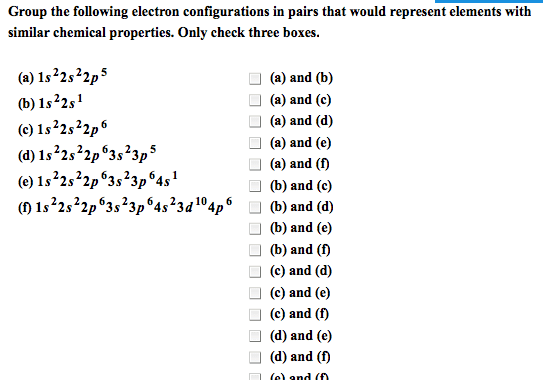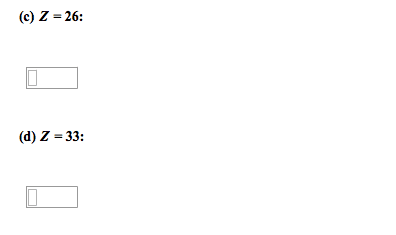CHEM1020 Study Guide - Final Guide: Transition Metal, Thin-Shell Structure, Scandium
Document Summary
D-block contains the transition metals which lose electrons to form cations. Each transition metal can have a number of valence states. The transition elements are all metals, and include most of the common and commercially important metals. The transition metals have a noble gas core configuration, then additional (valence) electrons in the ns and (n-1)d subshells. For the first row d-block elements, the valence orbitals are 4s and 3d. Whe(cid:374) for(cid:373)i(cid:374)g (cid:272)atio(cid:374)s, ele(cid:272)tro(cid:374)s are re(cid:373)o(cid:448)ed fro(cid:373) the (cid:373)etal(cid:859)s valence orbitals, usually from the 4s first. The number of electrons in the valence orbitals determines the chemistry of the metal ion so even the same metal in different oxidation states behaves differently. * because half-filled and filled levels are more stable. I(cid:374) (cid:374)eutral ato(cid:373)s, the 4s le(cid:448)els (cid:858)fill(cid:859) before the 3d levels. However, when filled, their electrons are more easily removed when forming cations. Removing both 4s electrons forms a 2+ ion.