7
answers
1
watching
172
views
rzansr2030Lv1
20 Oct 2023
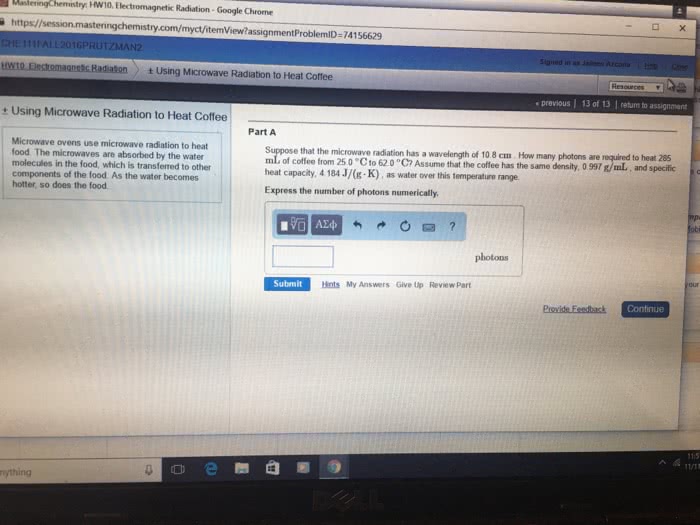
Suppose that the microwave radiation has a wavelength of 12 cmcm . How many photons are required to heat 265 mLmL of coffee from 25.0 ∘C∘C to 62.0 ∘C∘C? Assume that the coffee has the same density, 0.997 g/mLg/mL , and specific heat capacity, 4.184 J/(g⋅K)J/(g⋅K) , as water over this temperature range.
Suppose that the microwave radiation has a wavelength of 12 cmcm . How many photons are required to heat 265 mLmL of coffee from 25.0 ∘C∘C to 62.0 ∘C∘C? Assume that the coffee has the same density, 0.997 g/mLg/mL , and specific heat capacity, 4.184 J/(g⋅K)J/(g⋅K) , as water over this temperature range.
evangelistaLv10
12 Nov 2023
Already have an account? Log in
kankit11208Lv2
20 Oct 2023
Already have an account? Log in
Read by 1 person
oneclass0116Lv1
20 Oct 2023
Already have an account? Log in