5
answers
0
watching
258
views
cmall98Lv1
15 Jun 2023
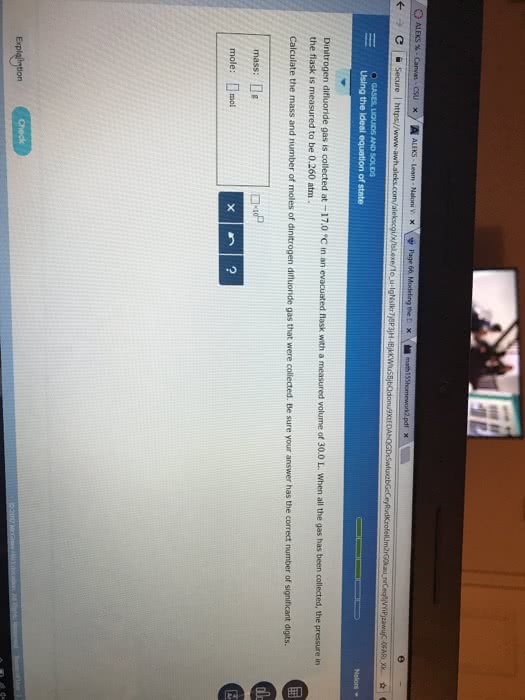
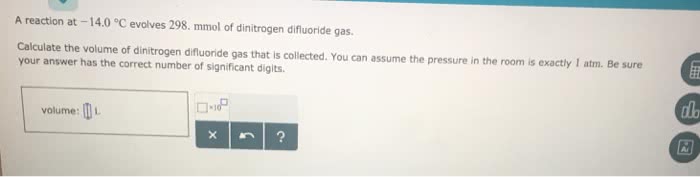
A reaction between liquid reactants takes place at 2.0°C in a sealed, evacuated vessel with a measured volume of 35.0L. Measurements show that the reaction produced 33.g of boron trifluoride gas.
Calculate the pressure of boron trifluoride gas in the reaction vessel after the reaction. You may ignore the volume of the liquid reactants. Round your answer to 2 significant digits.
A reaction between liquid reactants takes place at 2.0°C in a sealed, evacuated vessel with a measured volume of 35.0L. Measurements show that the reaction produced 33.g of boron trifluoride gas.
Calculate the pressure of boron trifluoride gas in the reaction vessel after the reaction. You may ignore the volume of the liquid reactants. Round your answer to 2 significant digits.
Liked by cmall98 and 1 others
15 Jun 2023
Read by 2 people
15 Jun 2023
Already have an account? Log in
Read by 2 people
Read by 2 people
15 Jun 2023
Already have an account? Log in