6
answers
0
watching
150
views
cmall98Lv1
15 Jun 2023
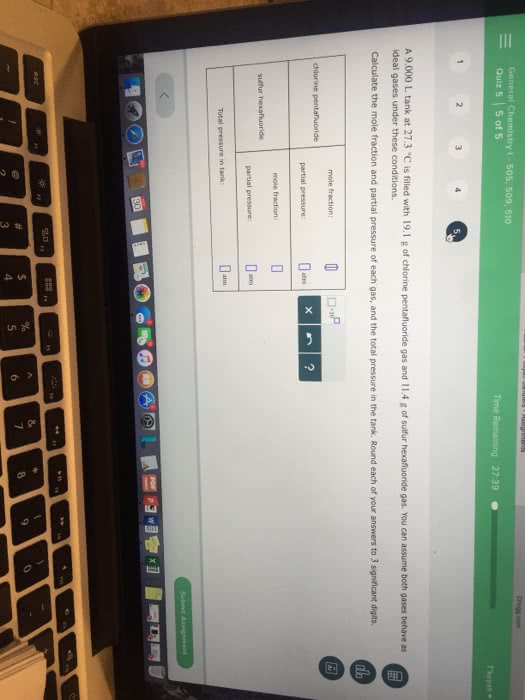
A 10.0L tank at 7.39°C is filled with 2.66g of dinitrogen monoxide gas and 19.6g of sulfur tetrafluoride gas. You can assume both gases behave as ideal gases under these conditions.
Calculate the total pressure in the tank. Round your answer to 3 significant digits.
A 10.0L tank at 7.39°C is filled with 2.66g of dinitrogen monoxide gas and 19.6g of sulfur tetrafluoride gas. You can assume both gases behave as ideal gases under these conditions.
Calculate the total pressure in the tank. Round your answer to 3 significant digits.
karimbalticLv7
3 Nov 2023
wahabmunir796Lv10
11 Jul 2023
Already have an account? Log in
22 Jun 2023
Already have an account? Log in
17 Jun 2023
Already have an account? Log in
Read by 1 person
15 Jun 2023
Already have an account? Log in