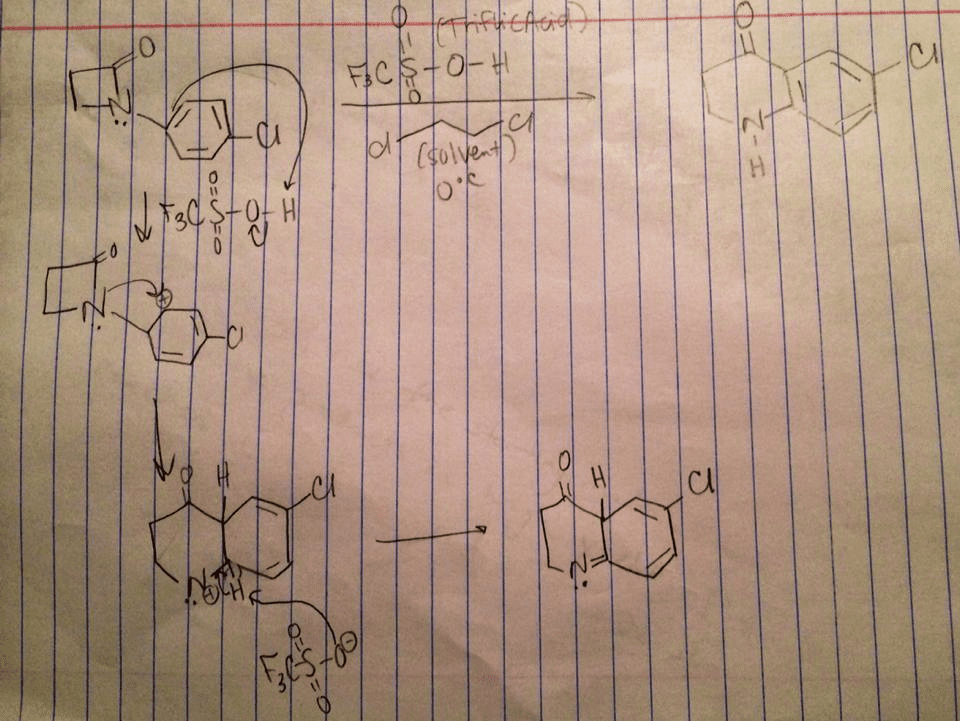
3
answers
1
watching
90
views
3
answers
1
watching
90
views
For unlimited access to Homework Help, a Homework+ subscription is required.
15 Feb 2023
Already have an account? Log in
15 Feb 2023
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Complete the following questions by using your classmates to answer the questions. You are allowed to walk around the classroom to help each other out and talk about the questions. You may also use your notes to help you check your answer or find answer.
|
If you dissolve a kool-aid packet into a pitcher of water is it a physical or chemical change?
Answer:
|
List 5 signs of chemical changes.
Answer:
|
If 1 had an object that has a density of 2.5g/ml and placed it into a container of water with a density of 1g/ml would the object sink or float?
Answer:
|
|
Draw an example of the atoms and molecules of a pure substance and a mixture.
Answer:
|
How can you tell if a liquid is more dense than another liquid?
Answer:
|
List 3 examples of physical change.
Answer:
|
|
Explain what happens to the mass of an object during a reaction.
Answer:
|
What are the units for:
Answer:
Graduated Cylinder: _____
Meter Stick: _____
Digital Scale: _____
|
If you tested your beverage with a piece of litmus paper and it turned red, do you have an acid or a base?
Answer:
|
|
What is the difference between soluble and insoluble?
Answer:
|
Give an example of a soluble solution and an insoluble solution.
Answer:
|
Explain the difference between chemical changes and physical changes?
Answer:
|
|
During our lab, we took vinegar and baking soda and combined them in a BEAKER. Why wasn't the mass before the reaction the same as the mass after? (what happened to the mass?)
Answer:
|
Why type of process do I use to separate a mixture? Physical or Chemical?
Answer:
|
How does the density of a copper penny compare to the density of a brick made out of copper and why? (Read this carefully)
Answer:
|
|
Explain how mass and volume are different?
Mass:
Volume:
|
Explain what density is?
Answer:
|
Give two characteristics of an acid and two characteristics of a base.
Answer:
|
|
What type of change occurs when water boils? Explain your answer.
Answer:
|
If you have an object with a volume of 10ml and a mass of 30g. What would its density be?
Answer:
|
I have 3 grams of vinegar and I mix it in a bag with 4 grams of baking soda. It starts to bubbles. Now what is the mass of the new substance?
Answer:
|
|
Explain the conservation of mass.
Answer:
|
Is burning something a chemical or physical change? Why?
Answer:
|
If I have an object with a density? of 10 and I cut the object into 4 what is the density of each piece? |