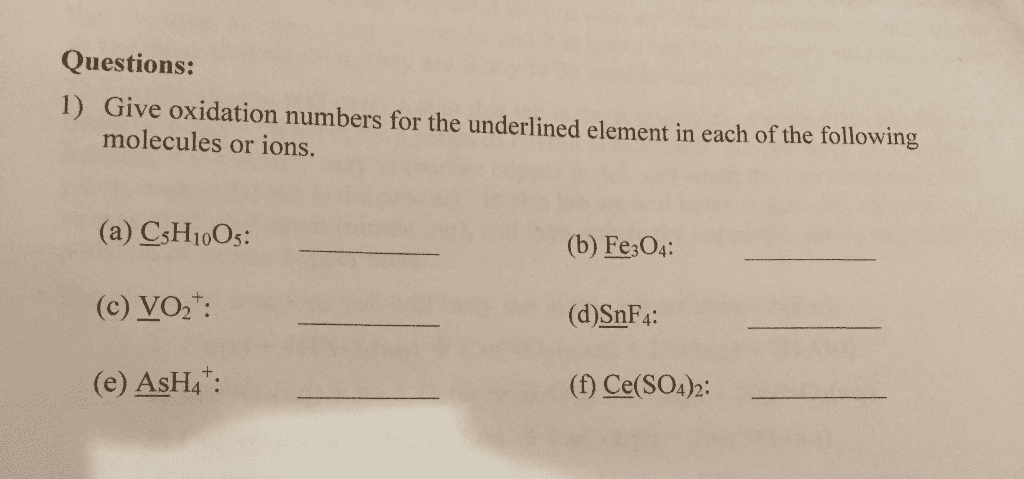2
answers
1
watching
378
views
applepieLv3
12 May 2022
- Give the oxidation number of the underlined atoms in the following molecules or ions:
- CH4
- C2H2
- ClF
- IF7
- C2H4
- K2CrO4
- K2Cr2O7
- NaHCO3
- NaIO3
- KO2
- Se8
- KAuCl4
- PtCl62-
- H5P3O10
- WO42-
- OF2
- Fe
- S2-
- ClO4-
- CaC2
- Give the oxidation number of the underlined atoms in the following molecules or ions:
- CH4
- C2H2
- ClF
- IF7
- C2H4
- K2CrO4
- K2Cr2O7
- NaHCO3
- NaIO3
- KO2
- Se8
- KAuCl4
- PtCl62-
- H5P3O10
- WO42-
- OF2
- Fe
- S2-
- ClO4-
- CaC2
Read by 7 people
alulerobertLv1
12 May 2022
Read by 8 people
12 May 2022
Already have an account? Log in