2
answers
1
watching
330
views
mx8hcvpd2fLv1
17 Dec 2021
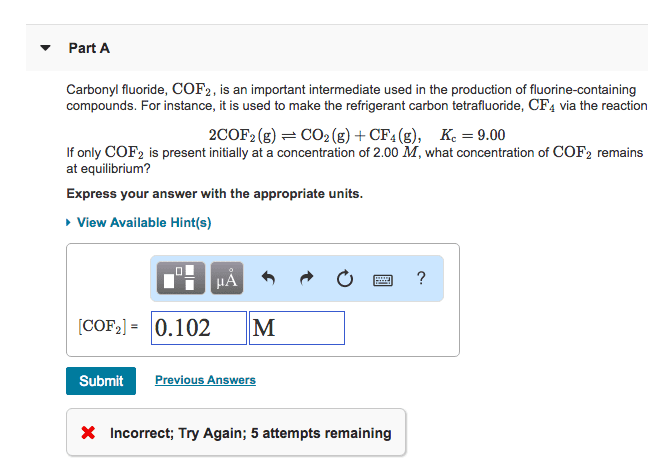
Carbonyl fluoride, COF2 , is an important intermediate used in the production of fluorine-containing compounds. For instance, it is used to make the refrigerant carbon tetrafluoride, CF4 via the reaction
2COF2(g)⇌CO2(g)+CF4(g), K=7.80 at 850 K
If only COF2 is present initially at a pressure of 244 kPa , what is the partial pressure of COF2 at equilibrium?
You can convert the units of pressure from kPa to bar , using the relation 1 kPa=0.01 bar.
Express your answer with the appropriate units.
Carbonyl fluoride, COF2 , is an important intermediate used in the production of fluorine-containing compounds. For instance, it is used to make the refrigerant carbon tetrafluoride, CF4 via the reaction
2COF2(g)⇌CO2(g)+CF4(g), K=7.80 at 850 K
If only COF2 is present initially at a pressure of 244 kPa , what is the partial pressure of COF2 at equilibrium?
You can convert the units of pressure from kPa to bar , using the relation 1 kPa=0.01 bar.
Express your answer with the appropriate units.
sherylwolvesLv4
19 Dec 2021
Read by 1 person