0
answers
0
watching
91
views
23 Nov 2019
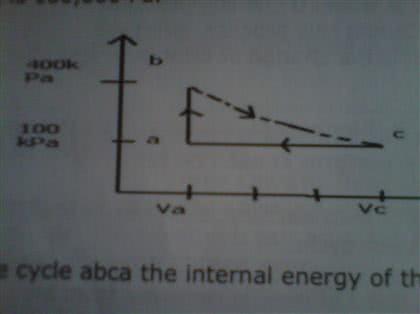
Figure 20-29 shows a reversible cycle through which 1.00 mole of amonatomic ideal gas is taken. Process bc is an adiabatic expansion,with pb = 14.0 atm and Vb = 3.60 x 10-3 m3. For the cycle, find (a)the energy added to the gas as heat, (b) the energy leaving the gasas heat, (c) the net work done by the gas, and (d) the efficiencyof the cycle.
Figure 20-29 shows a reversible cycle through which 1.00 mole of amonatomic ideal gas is taken. Process bc is an adiabatic expansion,with pb = 14.0 atm and Vb = 3.60 x 10-3 m3. For the cycle, find (a)the energy added to the gas as heat, (b) the energy leaving the gasas heat, (c) the net work done by the gas, and (d) the efficiencyof the cycle.