2
answers
0
watching
54
views
13 Dec 2019
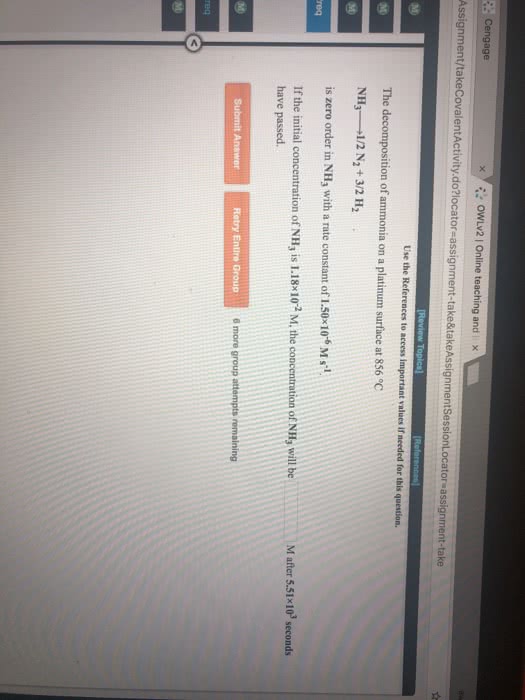
The decomposition of ammonia on a tungsten surface at 1100 °C
NH3-----.>1/2 N2 + 3/2 H2
is zero order in NH3.
In one experiment, when the initial concentration of NH3 was 1.49Ã10-2 M, the concentration of NH3 dropped to 2.68Ã10-3 M after 2.68Ã103 secondshad passed.
Based on these data, the rate constant for the reaction is M s-1.
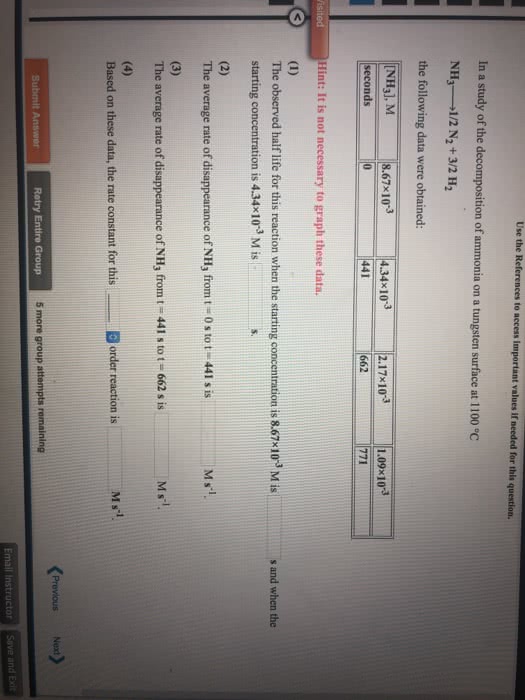
The decomposition of ammonia on a tungsten surface at 1100 °C
NH3-----.>1/2 N2 + 3/2 H2
is zero order in NH3.
In one experiment, when the initial concentration of NH3 was 1.49Ã10-2 M, the concentration of NH3 dropped to 2.68Ã10-3 M after 2.68Ã103 secondshad passed.
Based on these data, the rate constant for the reaction is M s-1.
jagdishkumarLv7
17 Jul 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Jarrod RobelLv2
17 Dec 2019
Get unlimited access
Already have an account? Log in