2
answers
0
watching
33
views
13 Dec 2019
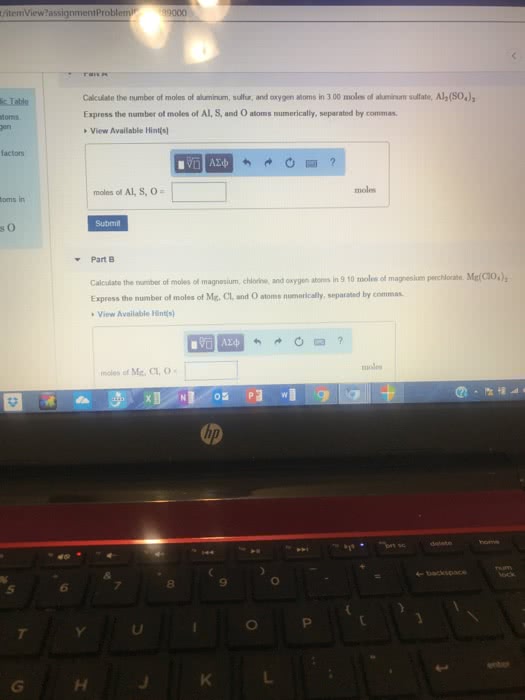
How many atoms are in 3.60 mol of pure aluminum?Use the periodic table to find the molar mass of aluminum. Use that and Avogadro\'s number to calculate the number of atoms of aluminum (Al).
How many atoms are in 3.60 mol of pure aluminum?Use the periodic table to find the molar mass of aluminum. Use that and Avogadro\'s number to calculate the number of atoms of aluminum (Al).
18 May 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Jean KeelingLv2
17 Dec 2019
Get unlimited access
Already have an account? Log in