2
answers
0
watching
13
views
12 Dec 2019
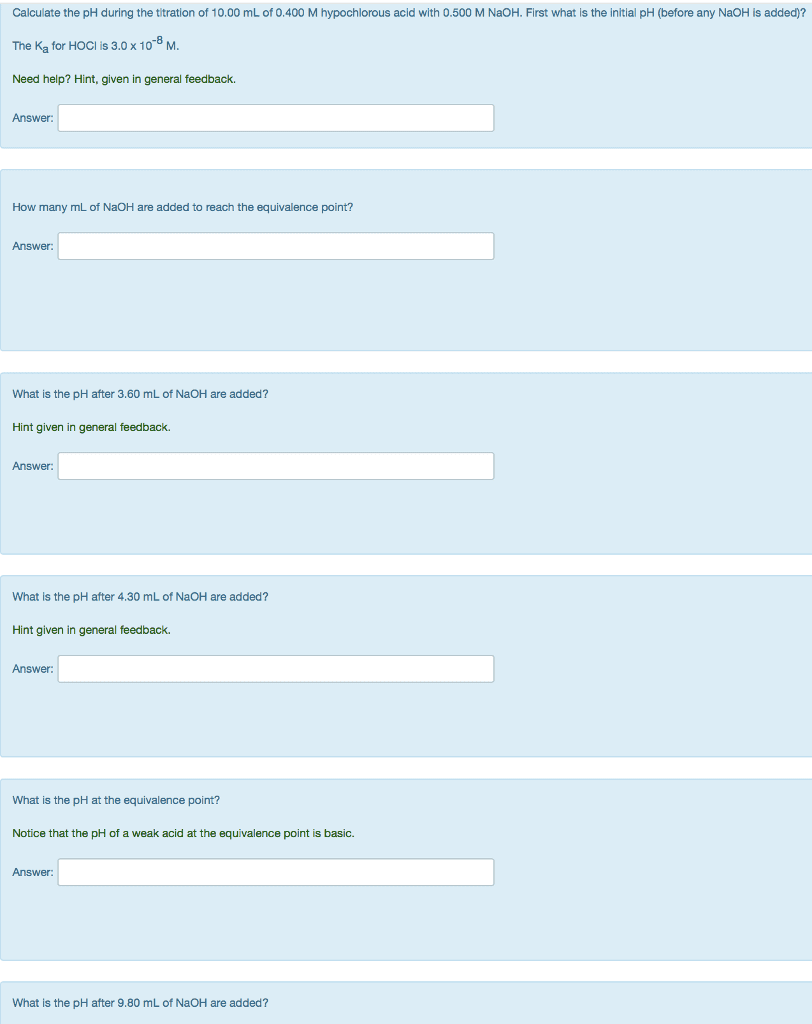
Titration Problem:
Lactic acid, HC3H5O3 (see right for structure), builds up in muscles after vigorous exercise. It is a weak acid with a pKa = 3.86.
Consider the following titration:
25.0 mL 0.500 M lactic acid is titrated with 1.00 M NaOH.
Answer the following questions:
1. What is the pH of the lactic acid solution before any NaOH is added?
2. What is the pH after 5.00 mL of 1.00 M NaOH is added?
3. What is the pH at the equivalence point?
4. What is the pH when 25.0 mL of 1.00 M NaOH has been added?
Titration Problem:
Lactic acid, HC3H5O3 (see right for structure), builds up in muscles after vigorous exercise. It is a weak acid with a pKa = 3.86.
Consider the following titration:
25.0 mL 0.500 M lactic acid is titrated with 1.00 M NaOH.
Answer the following questions:
1. What is the pH of the lactic acid solution before any NaOH is added?
2. What is the pH after 5.00 mL of 1.00 M NaOH is added?
3. What is the pH at the equivalence point?
4. What is the pH when 25.0 mL of 1.00 M NaOH has been added?
opn04125Lv3
14 Feb 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Jamar FerryLv2
13 Dec 2019
Get unlimited access
Already have an account? Log in