2
answers
0
watching
22
views
12 Dec 2019
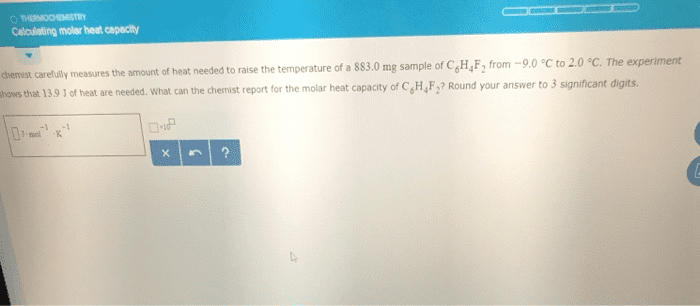
A chemist carefully measures the amount of heat needed to raise the temperature of a 895.0g sample of a pure substance from 6.1°C to 17.2°C . The experiment shows that 24.2kJ of heat are needed. What can the chemist report for the specific heat capacity of the substance? Round your answer to 3 significant digits.
A chemist carefully measures the amount of heat needed to raise the temperature of a 895.0g sample of a pure substance from 6.1°C to 17.2°C . The experiment shows that 24.2kJ of heat are needed. What can the chemist report for the specific heat capacity of the substance? Round your answer to 3 significant digits.
28 Jun 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Keith LeannonLv2
13 Dec 2019
Get unlimited access
Already have an account? Log in