2
answers
0
watching
29
views
11 Dec 2019
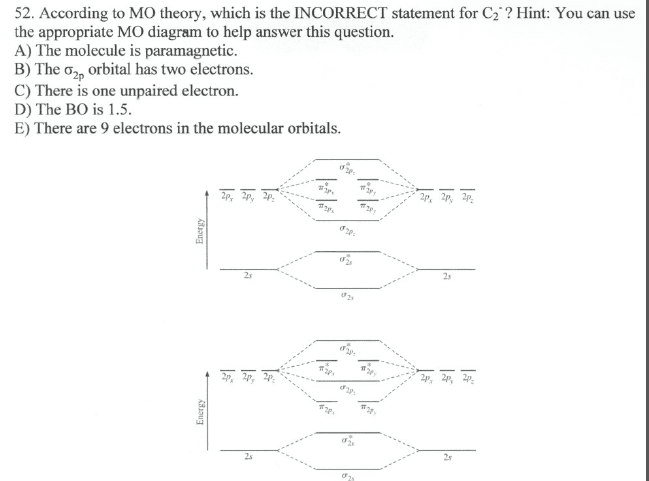
Which of the following statements are incorrect?
Select one:
a. Antibonding electrons are higher in energy than the atomic orbitals from which they came from.
b. At least two of the above are incorrect.
c. For the molecule NO, the molecular orbital model is preferred over the localized electron model because NO contains an unpaired electron.
d. Electrons in antibonding orbitals will cause a molecule to be paramagnetic.
e. According to the molecular orbital model, when bonding occurs between hydrogen and bromine to make HBr, the 1s orbital of the hydrogen atom no longer exists.
Which of the following statements are incorrect?
Select one:
a. Antibonding electrons are higher in energy than the atomic orbitals from which they came from.
b. At least two of the above are incorrect.
c. For the molecule NO, the molecular orbital model is preferred over the localized electron model because NO contains an unpaired electron.
d. Electrons in antibonding orbitals will cause a molecule to be paramagnetic.
e. According to the molecular orbital model, when bonding occurs between hydrogen and bromine to make HBr, the 1s orbital of the hydrogen atom no longer exists.
30 Apr 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Sixta KovacekLv2
13 Dec 2019
Get unlimited access
Already have an account? Log in