2
answers
0
watching
15
views
11 Dec 2019
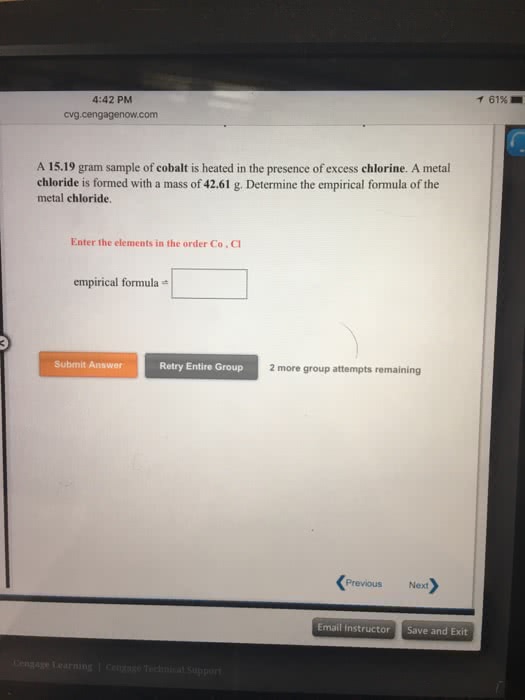
A 16.77 gram sample of iron is heated in the presence of excess chlorine. A metal chloride is formed with a mass of 38.05 g. Determine the empirical formula of the metal chloride.
A 16.77 gram sample of iron is heated in the presence of excess chlorine. A metal chloride is formed with a mass of 38.05 g. Determine the empirical formula of the metal chloride.
24 Feb 2024
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Beverley SmithLv2
13 Dec 2019
Get unlimited access
Already have an account? Log in