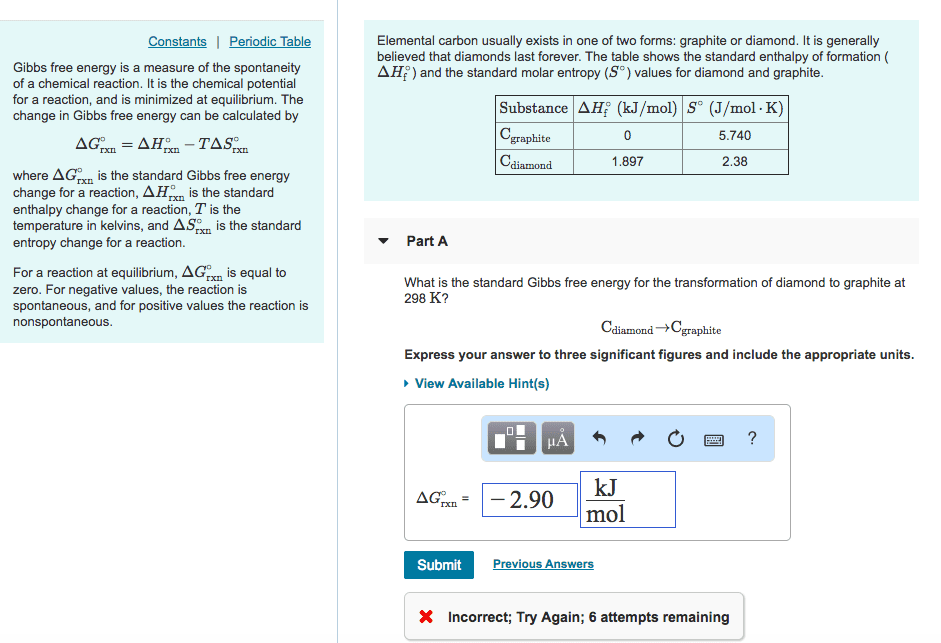
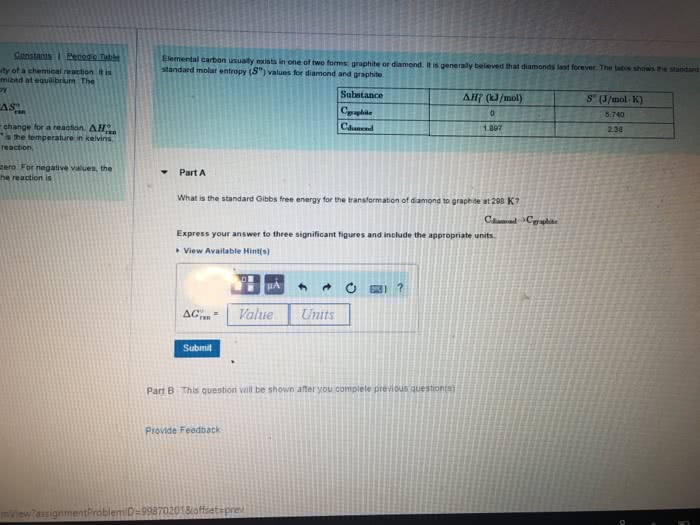
A) Suppose you dump a bucket of water (5.00kg at 15.0C) outside on a cold night where T = -10.0C, and the water spreads out over an area so large that the temperature of the ground remains at -10.0C, acting like an ideal âcold reservoir.â Calculate the entropy change of the water in going from 15.0C to -10.0C, and calculate the entropy change of the environment, absorbing heat while staying (approximately) at -10.0C. This problem requires you to use TWO equations for entropy change â of a substance changing temperature and for a substance with Q in or out but at constant T. I got somewhat less than 8000J/K for the âenvironmentâsâ âS.
B) Use your answers to part A to solve for the total entropy change of the UNIVERSE in that process (ie, add the entropy changes of all the parts, confirming that the entropy change of the UNIVERSE is positive.)
C) Some people uncomfortable with the idea of evolution try to argue against it because, they say, it says that more âorderedâ organisms evolve from less ordered organisms. Since entropy is a measure of âdisorder,â this canât happen. Now suppose for a moment that if you really did calculate the entropy of a bunch of âbiomassâ of 1 billion years ago, full of primitive organisms and a few inorganic compounds, and the equivalent âbiomassâ of a few people, and you found that the entropy WAS lower for the people, indicating that, yes indeed, the more complex organisms DO have lower entropy than the equivalent âprimitive stuffâ of which they were made. Why does this STILL not violate the second law of thermodynamics? (Hint: is part B fundamentally different from part A of this exercise, or are they fundamentally the same?)
A) Suppose you dump a bucket of water (5.00kg at 15.0C) outside on a cold night where T = -10.0C, and the water spreads out over an area so large that the temperature of the ground remains at -10.0C, acting like an ideal âcold reservoir.â Calculate the entropy change of the water in going from 15.0C to -10.0C, and calculate the entropy change of the environment, absorbing heat while staying (approximately) at -10.0C. This problem requires you to use TWO equations for entropy change â of a substance changing temperature and for a substance with Q in or out but at constant T. I got somewhat less than 8000J/K for the âenvironmentâsâ âS.
B) Use your answers to part A to solve for the total entropy change of the UNIVERSE in that process (ie, add the entropy changes of all the parts, confirming that the entropy change of the UNIVERSE is positive.)
C) Some people uncomfortable with the idea of evolution try to argue against it because, they say, it says that more âorderedâ organisms evolve from less ordered organisms. Since entropy is a measure of âdisorder,â this canât happen. Now suppose for a moment that if you really did calculate the entropy of a bunch of âbiomassâ of 1 billion years ago, full of primitive organisms and a few inorganic compounds, and the equivalent âbiomassâ of a few people, and you found that the entropy WAS lower for the people, indicating that, yes indeed, the more complex organisms DO have lower entropy than the equivalent âprimitive stuffâ of which they were made. Why does this STILL not violate the second law of thermodynamics? (Hint: is part B fundamentally different from part A of this exercise, or are they fundamentally the same?)