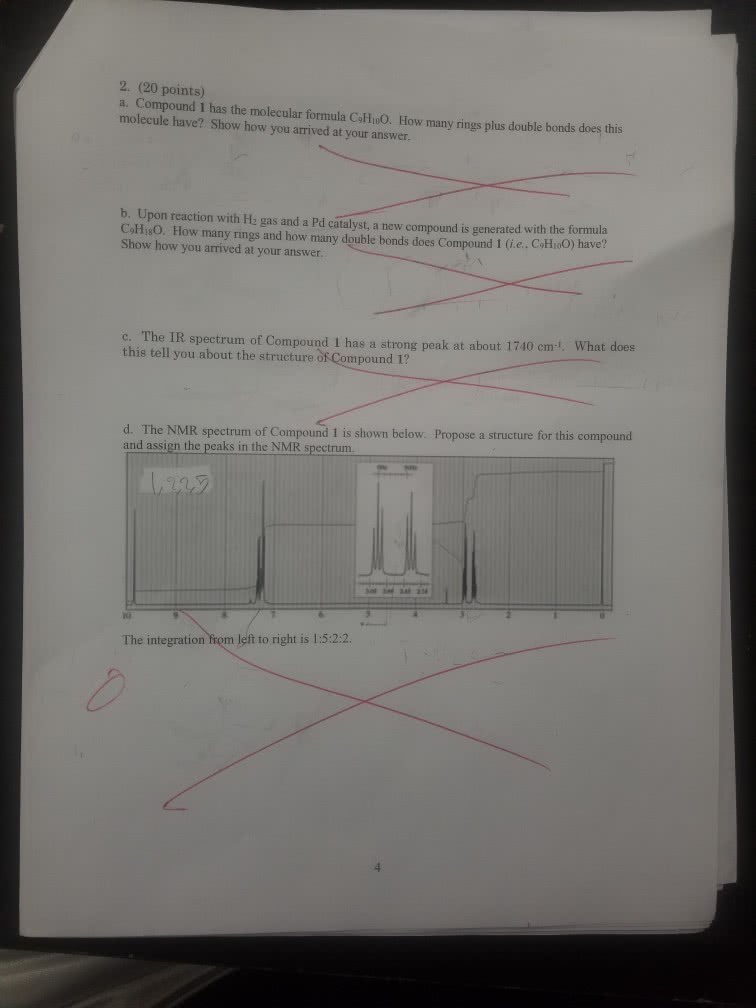
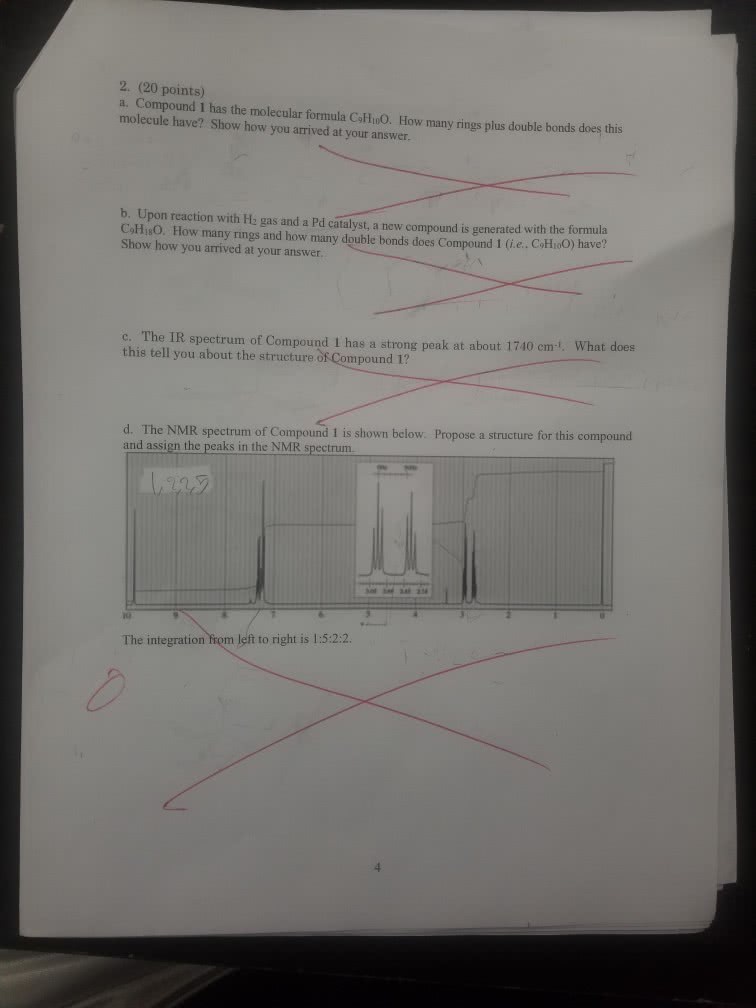
1
answer
1
watching
68
views
10 Mar 2022
(A)
A compound A with molecular formula C20H20ClBrFN5O3S is hydrogenated to give a compound B with molecular formula C20H36ClBrFN5O3S. How many rings does compound A have? Assume that sulfur in this case has a valency (not valence electrons) of 4 in these molecules and that all other atoms have standard valencies.
A. 4 B. 3 C. 2 D. 1 E. None of the above
(B)
How many total bonds does compound B have?â
A. 74 B. 73 C. 72 D. 71 E. None of the above
(A)
A compound A with molecular formula C20H20ClBrFN5O3S is hydrogenated to give a compound B with molecular formula C20H36ClBrFN5O3S. How many rings does compound A have? Assume that sulfur in this case has a valency (not valence electrons) of 4 in these molecules and that all other atoms have standard valencies.
A. 4 B. 3 C. 2 D. 1 E. None of the above
(B)
How many total bonds does compound B have?â
A. 74 B. 73 C. 72 D. 71 E. None of the above
12 Mar 2022
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in