2
answers
0
watching
27
views
11 Dec 2019
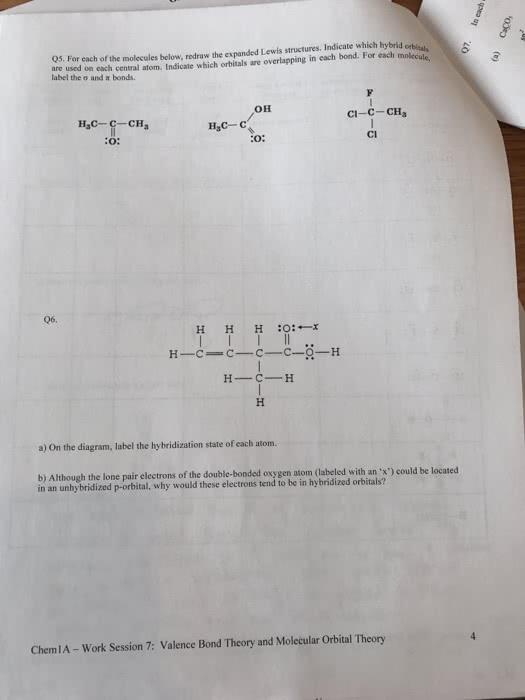
Please explain if the following are true or false and how you got your answer.
A. A molecule which contains polar bonds will always have a dipole moment.
B. Valence bond theory explains the bonding in diatomic molecules such as HCl without resortingto the use of hybrid orbitals.
C. In molecular orbital theory, combination of two atomic orbitals produces two molecular orbitals.
D. In applying molecular orbital theory, the bond order is calculated in the same way as with Lewis
structures.
E. In the valence bond treatment, overlap of an s orbital on one atom with an sp3 orbital
on another atom can give rise to a s bond
Please explain if the following are true or false and how you got your answer.
A. A molecule which contains polar bonds will always have a dipole moment.
B. Valence bond theory explains the bonding in diatomic molecules such as HCl without resortingto the use of hybrid orbitals.
C. In molecular orbital theory, combination of two atomic orbitals produces two molecular orbitals.
D. In applying molecular orbital theory, the bond order is calculated in the same way as with Lewis
structures.
E. In the valence bond treatment, overlap of an s orbital on one atom with an sp3 orbital
on another atom can give rise to a s bond
30 Apr 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Collen VonLv2
13 Dec 2019
Get unlimited access
Already have an account? Log in