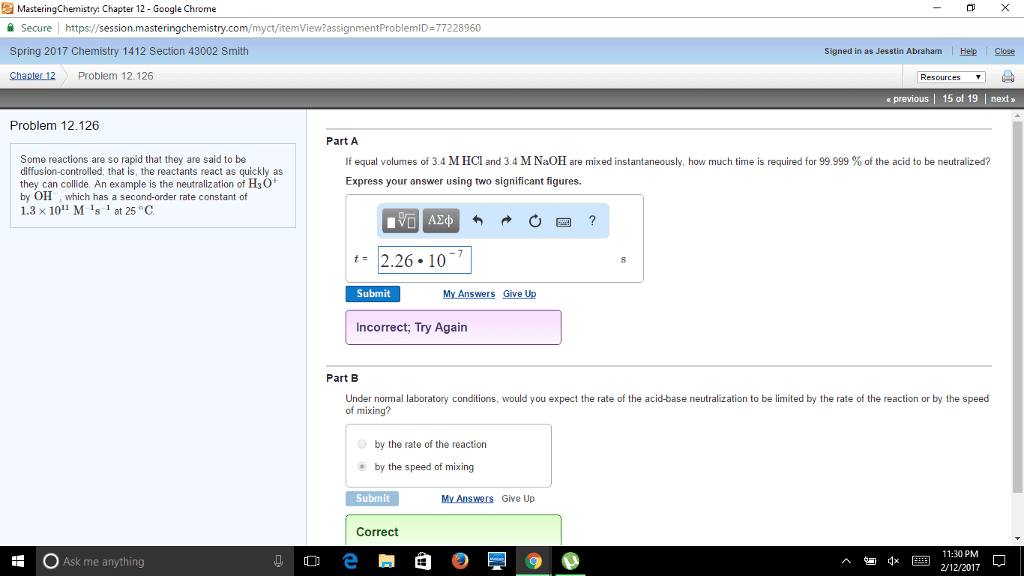
2
answers
0
watching
16
views
17 Nov 2019
Can I get help with these two problems please? I am at a total loss.
1. How much limestone in kilograms is required to completely neutralize a 4.6Ã109 L lake with a pH of 5.6?
2.What volume of HCl solution with a pH of 1.3 can be neutralized by 1 dose of milk of magnesia? (Assume two significant figures in your calculations.)
Can I get help with these two problems please? I am at a total loss.
1. How much limestone in kilograms is required to completely neutralize a 4.6Ã109 L lake with a pH of 5.6?
2.What volume of HCl solution with a pH of 1.3 can be neutralized by 1 dose of milk of magnesia? (Assume two significant figures in your calculations.)
sibinkarthikLv1
10 Oct 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Keith LeannonLv2
5 Nov 2019
Get unlimited access
Already have an account? Log in