2
answers
0
watching
52
views
17 Nov 2019


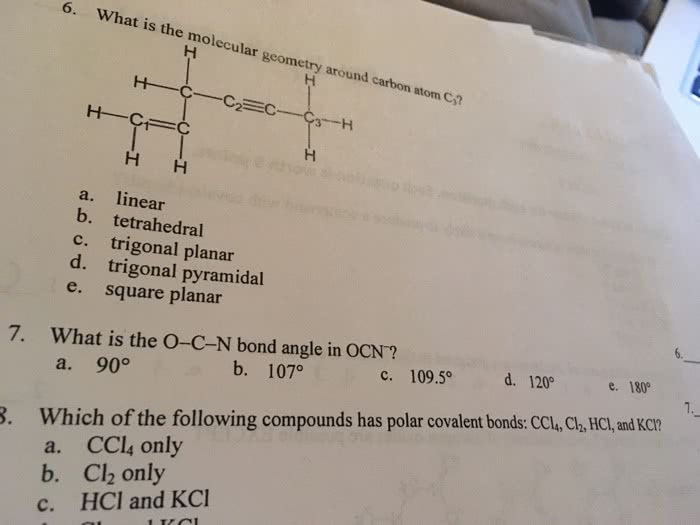
Which of the following bonds would be the least polar yet still be considered polar covalent Mg-O C-O O-O Si-O N-O What is the O-N-O bond angle in the nitrite ion? 90 degree 109 degree 120 degree 180 degree 180 degree and 90 degree The larger the difference in electronegativity between two bonded atoms, the more ionic the bond. the more covalent the bond. the more polar the bond. 1 only 2 only 3 only 1 and 3 only 2 and 3 only
2
answers
0
watching
52
views
For unlimited access to Homework Help, a Homework+ subscription is required.
18 Oct 2021
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Sixta KovacekLv2
9 Feb 2019
Get unlimited access
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232