2
answers
0
watching
15
views
17 Nov 2019
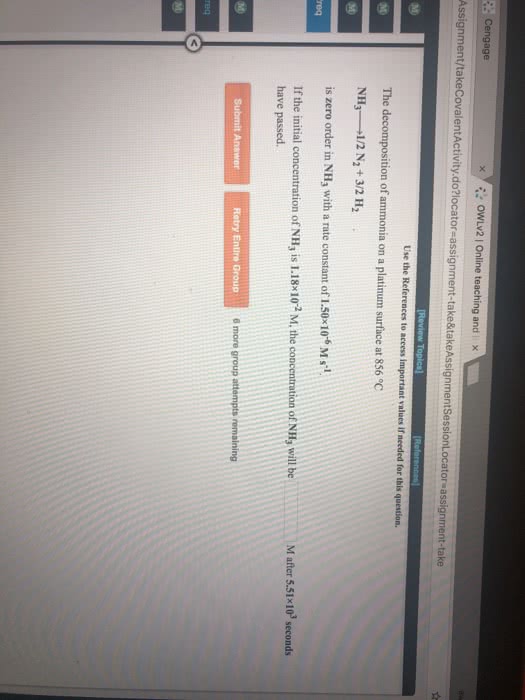
The rate constant for the zeroth-order decomposition of NH3 on a platinum surface at 856 âC is 1.50Ã10â6 M/s. How much time is required for the concentration of NH3 to drop from 6.60Ã10â3 M to 1.00Ã10â3 M?
The rate constant for the zeroth-order decomposition of NH3 on a platinum surface at 856 âC is 1.50Ã10â6 M/s. How much time is required for the concentration of NH3 to drop from 6.60Ã10â3 M to 1.00Ã10â3 M?
msmravicicLv2
26 Feb 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Casey DurganLv2
17 Nov 2019
Get unlimited access
Already have an account? Log in