2
answers
0
watching
16
views
17 Nov 2019
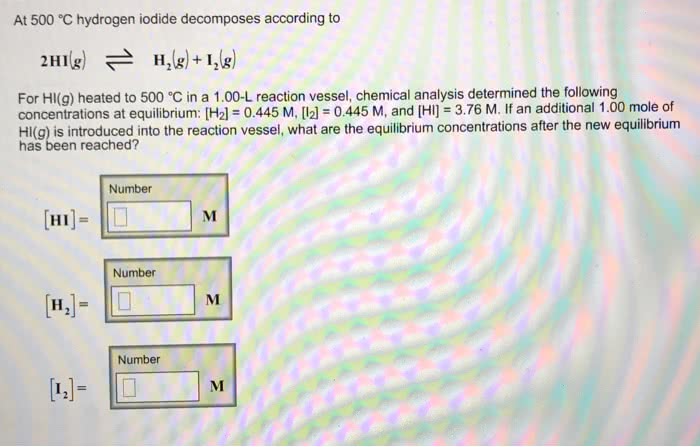
At 500 °C hydrogen iodide decomposes according to 2HI(g) arrows going back and forth H2(g)+ I2(g) For HI(g) heated to 500 °C in a 1.00-L reaction vessel, chemical analysis determined the following concentrations at equilibrium: [H2] = 0.382 M, [I2] = 0.382 M, and [HI] = 3.23 M. If an additional 1.00 mole of HI(g) is introduced into the reaction vessel, what are the equilibrium concentrations after the new equilibrium has been reached?
At 500 °C hydrogen iodide decomposes according to 2HI(g) arrows going back and forth H2(g)+ I2(g) For HI(g) heated to 500 °C in a 1.00-L reaction vessel, chemical analysis determined the following concentrations at equilibrium: [H2] = 0.382 M, [I2] = 0.382 M, and [HI] = 3.23 M. If an additional 1.00 mole of HI(g) is introduced into the reaction vessel, what are the equilibrium concentrations after the new equilibrium has been reached?
12 Feb 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Nelly StrackeLv2
24 Jan 2019
Get unlimited access
Already have an account? Log in