2
answers
0
watching
20
views
17 Nov 2019
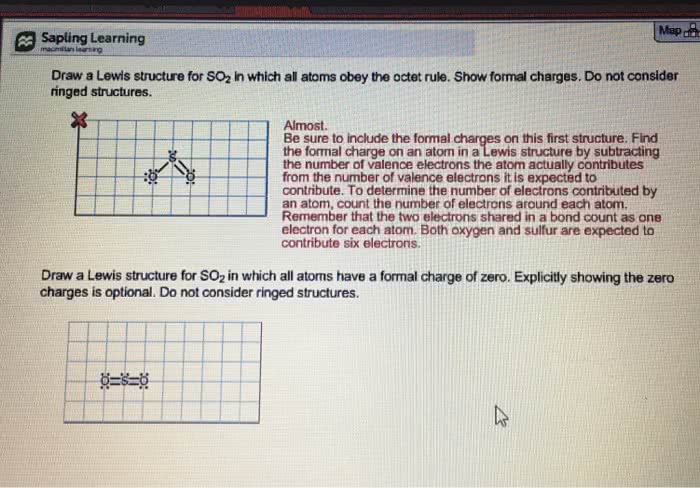
Phosphoryl Chloride, POCl3, is a reagent that is used to make phosphate triesters from alcohols. (A) Draw a Lewis Dot structure that satisfies the octet rule for each atom in POCl3 then calculate the formal charge for each atom in the structure. (B) Draw a resonance structure of POCl3 in which all the atoms have a formal charge of zero.
Phosphoryl Chloride, POCl3, is a reagent that is used to make phosphate triesters from alcohols. (A) Draw a Lewis Dot structure that satisfies the octet rule for each atom in POCl3 then calculate the formal charge for each atom in the structure. (B) Draw a resonance structure of POCl3 in which all the atoms have a formal charge of zero.
shubham20Lv2
6 Nov 2022
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Casey DurganLv2
27 Apr 2019
Get unlimited access
Already have an account? Log in