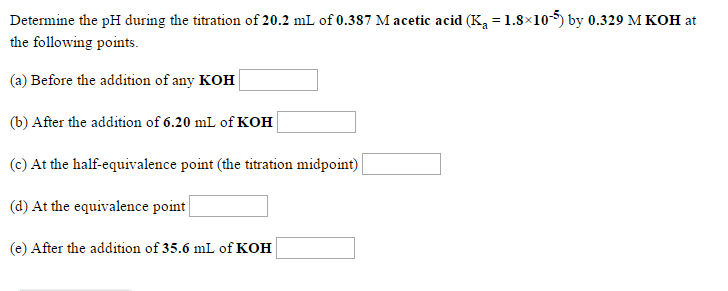
3
answers
1
watching
253
views
23 Jul 2021
A 25.0mL of 0.250M acetic acid (CH3COOH) is titrated with 0.10M KOH. Calculate the pH after the addition of the following amounts of KOH
a) 0 mL b) 61 mL c) at the equivalence point d) 65 mL
A 25.0mL of 0.250M acetic acid (CH3COOH) is titrated with 0.10M KOH. Calculate the pH after the addition of the following amounts of KOH
a) 0 mL b) 61 mL c) at the equivalence point d) 65 mL
chemosmart-007Lv10
23 Jul 2021
Already have an account? Log in
John Edward CayasLv10
23 Jul 2021
Already have an account? Log in