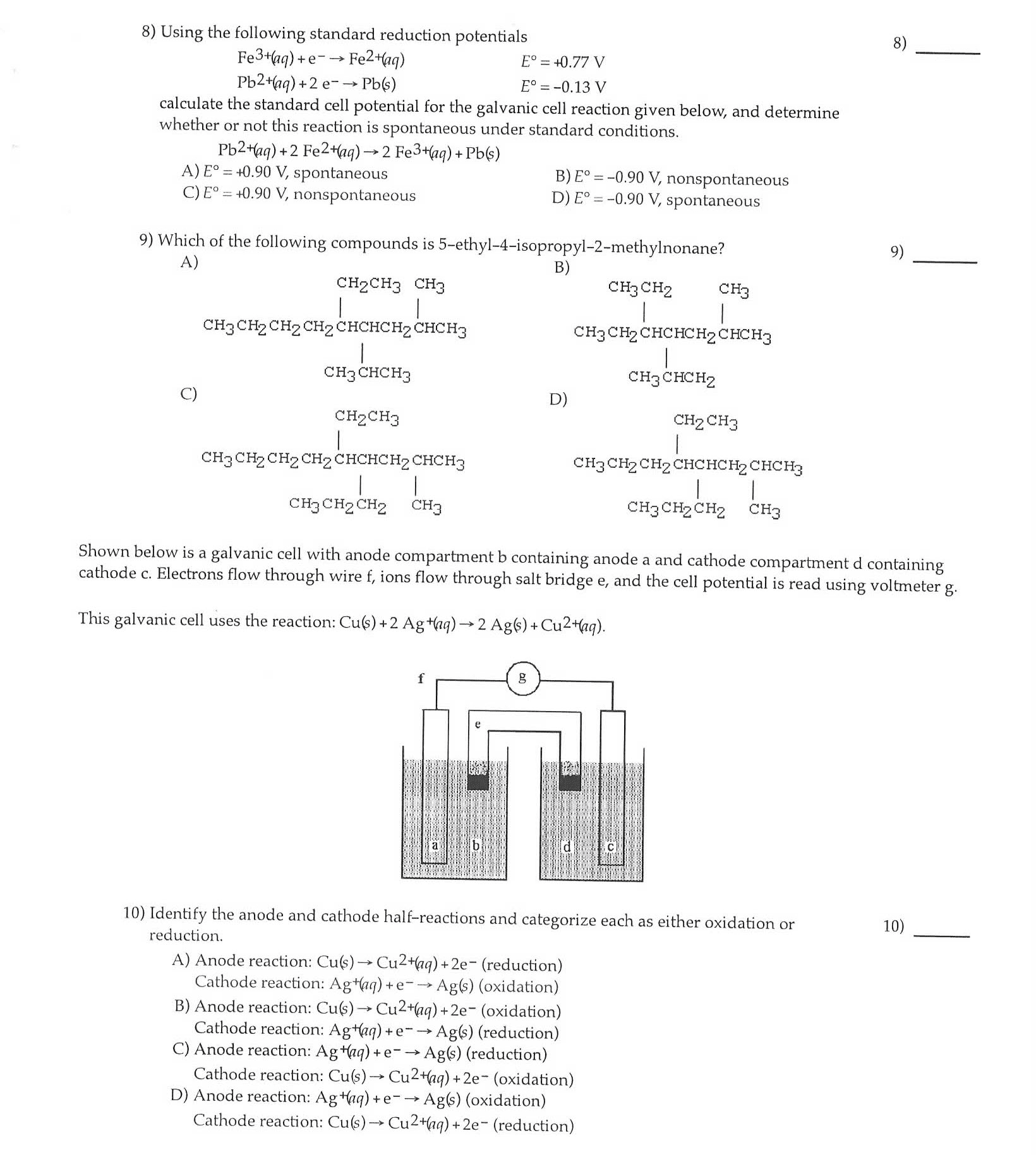
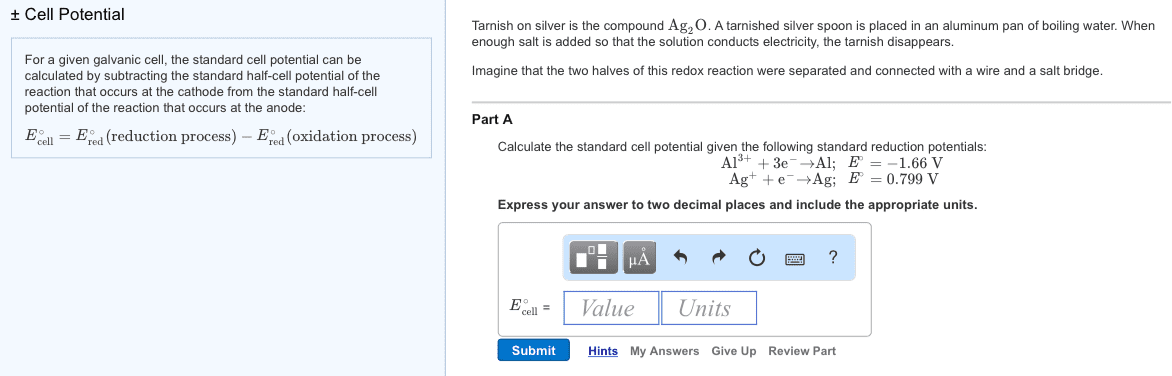
2
answers
1
watching
246
views
amberwasp281Lv1
20 Apr 2021
A certain half-reaction has a standard reduction potential E0red=+0.99V. An engineer proposes using this half-reaction at the cathode of a galvanic cell that must provide at least 1.30V of electrical power. The cell will operate under standard conditions. Note for advanced students: assume the engineer requires this half-reaction to happen at the cathode of the cell.
A certain half-reaction has a standard reduction potential E0red=+0.99V. An engineer proposes using this half-reaction at the cathode of a galvanic cell that must provide at least 1.30V of electrical power. The cell will operate under standard conditions. Note for advanced students: assume the engineer requires this half-reaction to happen at the cathode of the cell.
2
answers
1
watching
246
views
For unlimited access to Homework Help, a Homework+ subscription is required.
John Edward CayasLv10
20 Apr 2021
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232