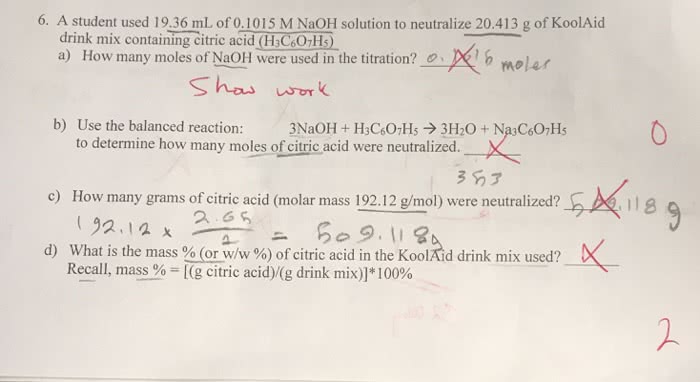2
answers
0
watching
365
views
14 Apr 2021
A student performs a titration to determine the amount of citric acid in apple juice. A 20.0 mL sample of this juice requires 12.84 mL of a standard NaOH solution, with a concentration of 0.1015 M.
Express the volume of NaOH solution in liters.
Calculate the number of moles of OH- required for the titration.
How many moles of H+ were titrated?
Calculate the number of moles of citric acid were titrated.
Calculate the mass of citric acid in the sample (in grams).
Calculate the concentration of citric acid in the juice (in grams/liter).
A student performs a titration to determine the amount of citric acid in apple juice. A 20.0 mL sample of this juice requires 12.84 mL of a standard NaOH solution, with a concentration of 0.1015 M.
Express the volume of NaOH solution in liters.
Calculate the number of moles of OH- required for the titration.
How many moles of H+ were titrated?
Calculate the number of moles of citric acid were titrated.
Calculate the mass of citric acid in the sample (in grams).
Calculate the concentration of citric acid in the juice (in grams/liter).
Read by 3 people