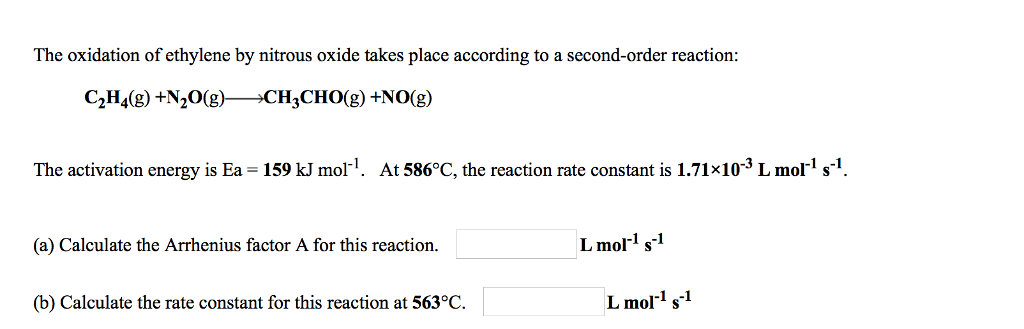2
answers
2
watching
634
views
azurepug516Lv1
2 Mar 2021
- 1.The experimental rate law of a simple reaction A → B + C is v = k[A]. (a) Determine total order of the reaction. (b) Imagine initial conditions at which the rate equals v0. Calculate the change in the reaction rate that will be observed when the concentration of A will be tripled.
2.Calculate the activation energy for a first-order reaction if its rate constant increases from 10 s–1 to 100 s–1 as the temperature increases from 300 to 330 K.
3.Consider a simple first-order reaction A → B. If you start with solution of 0.1 mol/L A, it takes 20 s to go to a concentration of 0.05 mol/L of A. (a) What is the value of the rate constant for this reaction? (b) Calculate half-life of the reaction.
- 1.The experimental rate law of a simple reaction A → B + C is v = k[A]. (a) Determine total order of the reaction. (b) Imagine initial conditions at which the rate equals v0. Calculate the change in the reaction rate that will be observed when the concentration of A will be tripled.
2.Calculate the activation energy for a first-order reaction if its rate constant increases from 10 s–1 to 100 s–1 as the temperature increases from 300 to 330 K.
3.Consider a simple first-order reaction A → B. If you start with solution of 0.1 mol/L A, it takes 20 s to go to a concentration of 0.05 mol/L of A. (a) What is the value of the rate constant for this reaction? (b) Calculate half-life of the reaction.
Liked by oluwatosinayenin and 1 others
2 Jun 2021
jddjdjsjsjdLv1
25 Feb 2024
Already have an account? Log in