2
answers
2
watching
1,412
views
tealdog287Lv1
2 Mar 2021
Li2SO4(aq)+Sr(ClO3)2(aq) → SrSO4(s)+2LiClO3(aq)
Li2SO4(aq)+Sr(ClO3)2(aq) → SrSO4(s)+2LiClO3(aq)
The molecular equation you determined in Part B is shown above for your convenience. Examine each of the chemical species involved to determine the ions that would be present in solution.
Be sure to consider both the coefficients and subscripts of the molecular equation, and then write this precipitation reaction in the form of a balanced complete ionic equation.
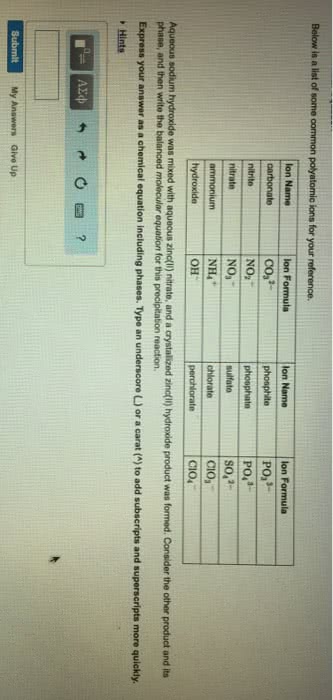
Express your answer as a chemical equation including phases. Type an underscore (_) or a carat (^) to add subscripts and superscripts more quickly.
Li2SO4(aq)+Sr(ClO3)2(aq) → SrSO4(s)+2LiClO3(aq)
Li2SO4(aq)+Sr(ClO3)2(aq) → SrSO4(s)+2LiClO3(aq)
The molecular equation you determined in Part B is shown above for your convenience. Examine each of the chemical species involved to determine the ions that would be present in solution.
Be sure to consider both the coefficients and subscripts of the molecular equation, and then write this precipitation reaction in the form of a balanced complete ionic equation.
Express your answer as a chemical equation including phases. Type an underscore (_) or a carat (^) to add subscripts and superscripts more quickly.
Liked by hannah.gonzalez.st60 and 2 others
2 Jun 2021
16 Mar 2024
Already have an account? Log in