2
answers
0
watching
337
views
19 Jan 2021
The freezing point of water is 0.00°C at 1 atmosphere.
How many grams of cobalt(II) chloride (129.8 g/mol), must be dissolved in 228.0 grams of water to reduce the freezing point by 0.450°C ? Refer to the table below for the necessary boiling or freezing point constant.
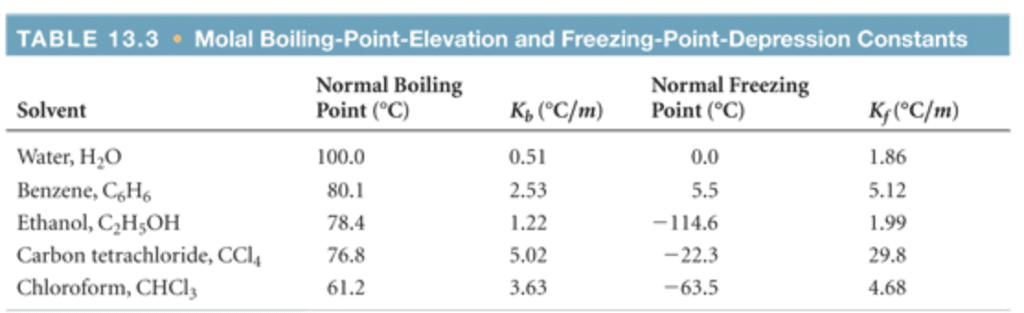
Solvent
Formula
Kb (°C/m)
Kf (°C/m)
Water
H2O
0.512
1.86
Ethanol
CH3CH2OH
1.22
1.99
Chloroform
CHCl3
3.67
Benzene
C6H6
2.53
5.12
Diethyl ether
CH3CH2OCH2CH3
2.02
The freezing point of water is 0.00°C at 1 atmosphere.
How many grams of cobalt(II) chloride (129.8 g/mol), must be dissolved in 228.0 grams of water to reduce the freezing point by 0.450°C ? Refer to the table below for the necessary boiling or freezing point constant.
| Solvent | Formula | Kb (°C/m) | Kf (°C/m) |
|---|---|---|---|
| Water | H2O | 0.512 | 1.86 |
| Ethanol | CH3CH2OH | 1.22 | 1.99 |
| Chloroform | CHCl3 | 3.67 | |
| Benzene | C6H6 | 2.53 | 5.12 |
| Diethyl ether | CH3CH2OCH2CH3 | 2.02 |
2 Jun 2021
Already have an account? Log in