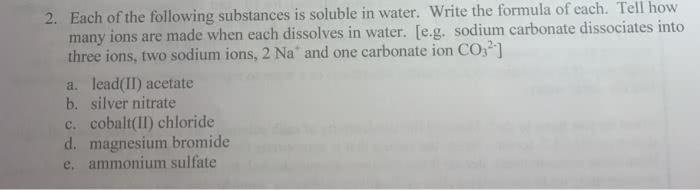1
answer
0
watching
194
views
28 Nov 2020
In the compound MgCl2, the subscript 2 indicates that:
- a) there are two magnesium ions for each ion of chlorine
- b) the chloride ion is twice the size of the magnesium ion
- c) magnesium and chlorine to form a double covalent bond
- d) there are two chloride ions for each magnesium ion
In the compound MgCl2, the subscript 2 indicates that:
- a) there are two magnesium ions for each ion of chlorine
- b) the chloride ion is twice the size of the magnesium ion
- c) magnesium and chlorine to form a double covalent bond
- d) there are two chloride ions for each magnesium ion
Amarildo KociajLv10
24 Jan 2021