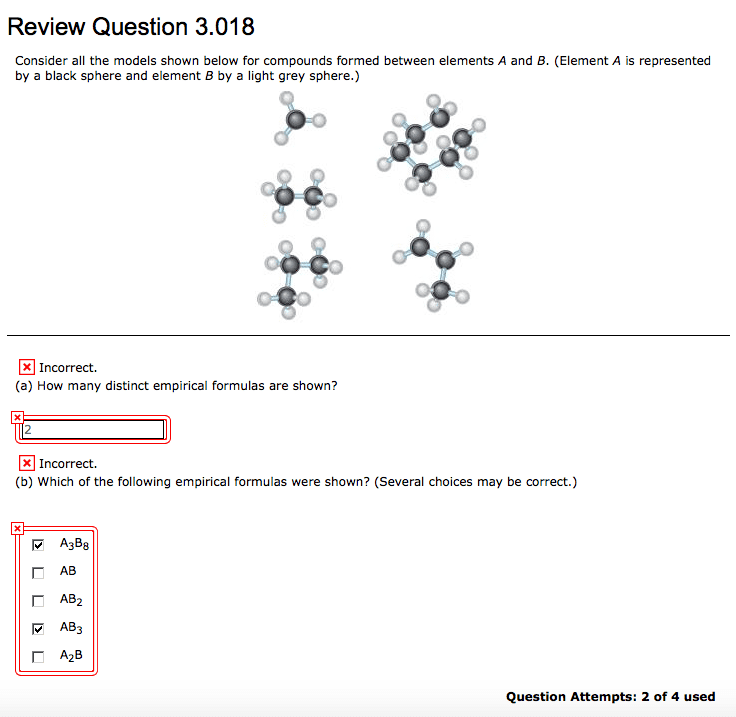
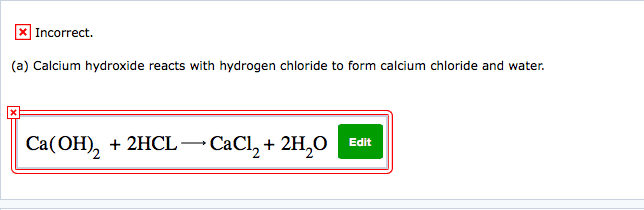
1
answer
0
watching
143
views
28 Nov 2020
a) Calculate the molar mass of ammonium nitrate(NH4NO3)? b) What is the mass percentage of Nitrogen in ammonium nitrate? c) How many kilograms of Nitrogen is present in a 10.0 lb bag of ammonium nitrate? d) How many kilograms of ammonium nitrate contains 1.0 lb of nitrogen? (1 lb = 453.6 g)
a) Calculate the molar mass of ammonium nitrate(NH4NO3)? b) What is the mass percentage of Nitrogen in ammonium nitrate? c) How many kilograms of Nitrogen is present in a 10.0 lb bag of ammonium nitrate? d) How many kilograms of ammonium nitrate contains 1.0 lb of nitrogen? (1 lb = 453.6 g)
Kottherva SreevidyaLv10
8 Jan 2021