1
answer
0
watching
214
views
goldlouse59Lv1
28 Nov 2020
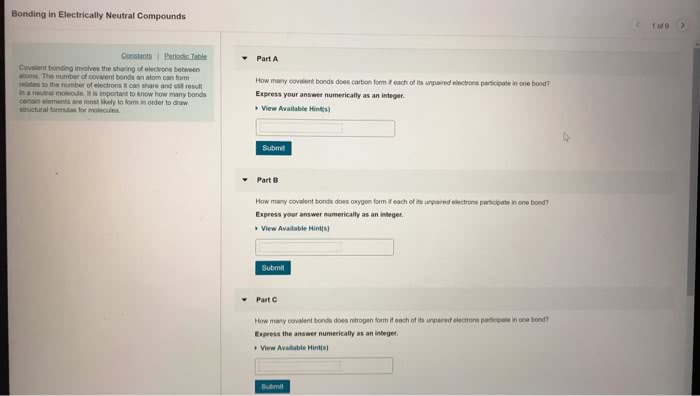
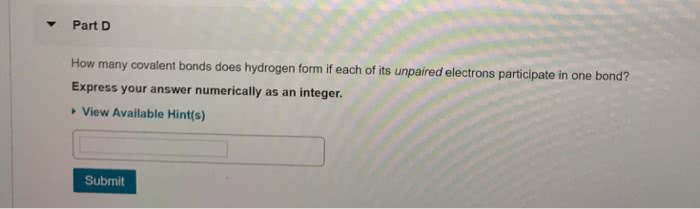
What is the fundamental difference between covalent and ionic bonding?
(a) Covalent bonding involves only the outermost electron shell; ionic bonding also involves the next electron shell inside the outermost shell.
(b) Covalent bonds form between atoms of the same element; ionic bonds form between atoms of different elements.
(c) In a covalent, the partners share a pair of electrons; in an ionic bond, one partner accepts electrons from the other.
(d) In covalent bonding, both partners end up with filled outer electron shells; in ionic bonding, one partner does and the other does not.
What is the fundamental difference between covalent and ionic bonding?
(a) Covalent bonding involves only the outermost electron shell; ionic bonding also involves the next electron shell inside the outermost shell.
(b) Covalent bonds form between atoms of the same element; ionic bonds form between atoms of different elements.
(c) In a covalent, the partners share a pair of electrons; in an ionic bond, one partner accepts electrons from the other.
(d) In covalent bonding, both partners end up with filled outer electron shells; in ionic bonding, one partner does and the other does not.
Brian HingpitLv10
8 Jan 2021