1
answer
0
watching
251
views
blackyak386Lv1
28 Nov 2020
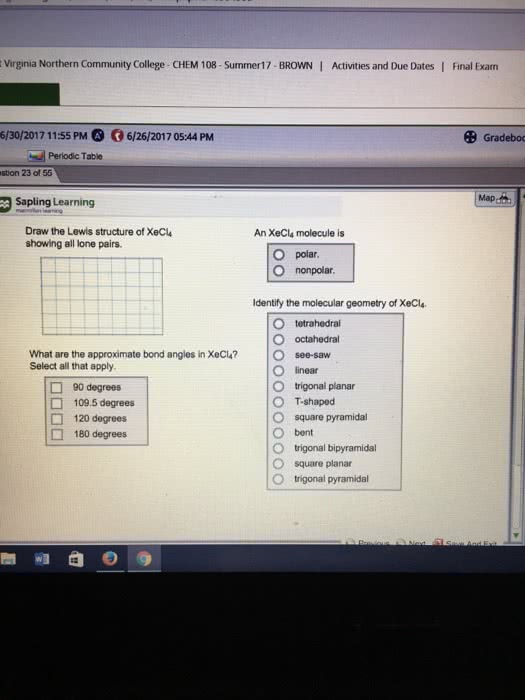
Draw the Lewis structure of 
Identify the molecular geometry of 
T‑shaped
square pyramidal
bent
linear
trigonal pyramidal
octahedral
tetrahedral
trigonal planar
trigonal bipyramidal
see‑saw
square planar
What is the hybridization of the central atom?








Draw the Lewis structure of 
Identify the molecular geometry of 
T‑shaped
square pyramidal
bent
linear
trigonal pyramidal
octahedral
tetrahedral
trigonal planar
trigonal bipyramidal
see‑saw
square planar
What is the hybridization of the central atom?










What are the ideal bond angles of this geometry?




90°
120°
An 

polar.
nonpolar.
Verified Answer
Allen DinoLv9
23 Jan 2021
28 Jan 2021
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.