1
answer
1
watching
1,038
views
limemule720Lv1
10 Oct 2020
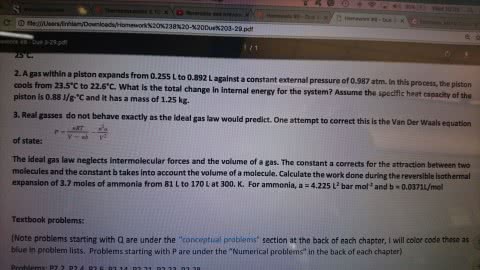
A sample consisting of 1.00 mol of a van der Waals gas is compressed from 20.0 dm3 to 10.0 dm3 at 300 K. In the process, 20.2 kJ of work is done on the gas. Given that µ = {(2a/RT) − b}/Cp,m, with Cp,m = 38.4 J K−1 mol−1 , a = 3.60 dm6 atm mol−2 , and b = 0.44 dm3 mol−1 , calculate ∆H for the process.
A sample consisting of 1.00 mol of a van der Waals gas is compressed from 20.0 dm3 to 10.0 dm3 at 300 K. In the process, 20.2 kJ of work is done on the gas. Given that µ = {(2a/RT) − b}/Cp,m, with Cp,m = 38.4 J K−1 mol−1 , a = 3.60 dm6 atm mol−2 , and b = 0.44 dm3 mol−1 , calculate ∆H for the process.
Liked by dhiren20052003
Robert KubaraLv10
11 Feb 2021