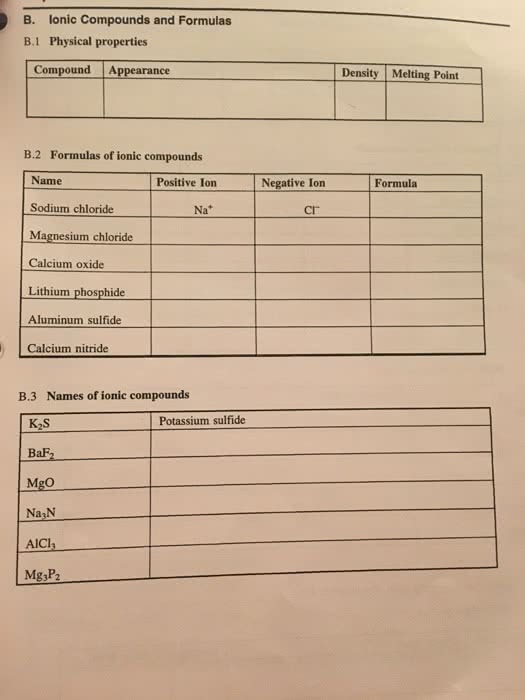
1
answer
0
watching
231
views
6 Oct 2020
What is the chemical formula for the following binary ionic compounds?
a.) Gallium nitride
b.) Zinc chloride
c.) Magnesium sulfide
d.) Aluminum nitride
What is the chemical formula for the following binary ionic compounds?
a.) Gallium nitride
b.) Zinc chloride
c.) Magnesium sulfide
d.) Aluminum nitride
1
answer
0
watching
231
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Matt OcampoLv10
19 Nov 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Write the Formula/ Formula Unit for the following Compounds
Determining the formula for Magnesium Fluoride?
- identify the charges

- Cross the Charges,
- If the subscript is a 1 it does not need to be written.
- If there is a common subscript such as 2 as in


Write Formula Unit for the Below Ionic Compounds
|
|
Name |
Cation (+) |
Anion (-) |
Formula |
|
1 |
Sodium chloride |
|||
|
2 |
Aluminum chloride |
|||
|
3 |
Aluminum phosphide |
|||
|
4 |
Magnesium oxide |
|||
|
5 |
Cesium fluoride |
|||
|
6 |
Strontium nitride |
|||
|
7 |
Lithium sulfide |
|||
|
8 |
Calcium chloride |
|||
|
9 |
Sodium bromide |
|||
|
10 |
Beryllium iodide |
|||
|
11 |
Strontium fluoride |
|||
|
12 |
Aluminum fluoride |
|||
|
13 |
Potassium nitride |
|||
|
14 |
Sodium sulfide |
|||
|
15 |
Lithium oxide |
|||
|
16 |
Calcium oxide |