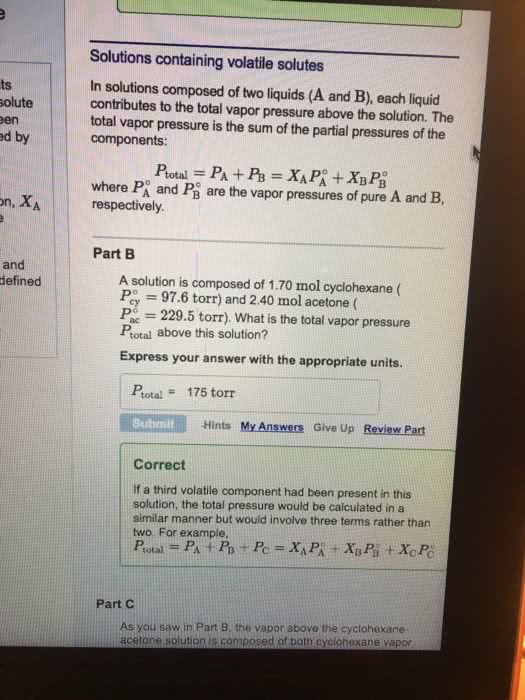
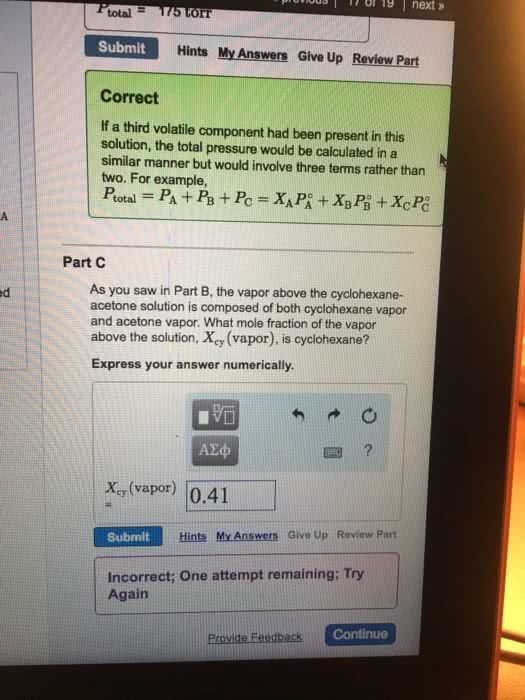
A liquid solution consists of 0.30 mole fraction ethylene dibromide, C2H4Br2, and 0.70 mole fraction propylene dibromide, C3H6Br2. Both ethylene dibromide and propylene dibromide are volatile liquids; their vapor pressures at 85°C are 173 mmHg and 127 mmHg, respectively. Assume that each compound follows Raoult’s law in the solution. Calculate the total vapor pressure of the solution.
A liquid solution consists of 0.30 mole fraction ethylene dibromide, C2H4Br2, and 0.70 mole fraction propylene dibromide, C3H6Br2. Both ethylene dibromide and propylene dibromide are volatile liquids; their vapor pressures at 85°C are 173 mmHg and 127 mmHg, respectively. Assume that each compound follows Raoult’s law in the solution. Calculate the total vapor pressure of the solution.
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
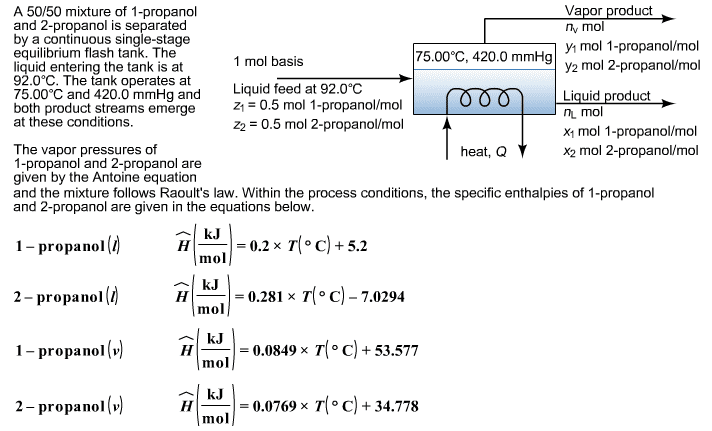
A pending refinery operation is being designed. A gas stream in this operation is to be partially condensed at 80. The composition of the gas is:
A: 5% (mole)
B: 30%
C: 55%
D: 10%
Determine the pressure for the partial condenser required to condense 75% of the gas stream. Assume Raoultâs law is valid. The Antoine equation and the appropriate coefficients are given below:
with in mmHg and T in .
| ai | bi | ci | |
| A | 6.6783 | 1190.0342 | 245.8321 |
| B | 6.8991 | 1245.0565 | 233.0989 |
| C | 7.0124 | 1309.9879 | 220.8011 |
| D | 7.2001 | 1469.9987 | 212.8345 |
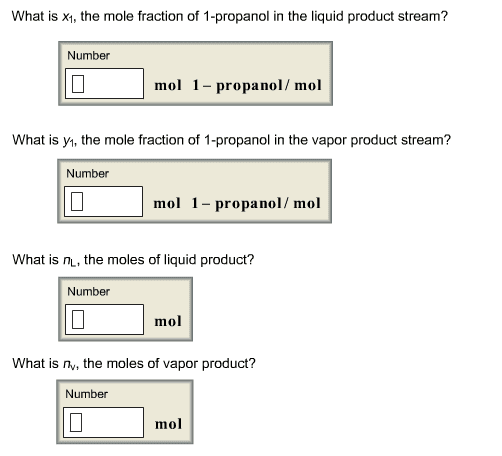
Use Excel to do the following:
Calculate the vapor pressures of the pure components.
Assume 100 moles of feed gas and calculate the moles of vapor and liquid that must come from the condenser.
Write material balances for each component around the condenser.
Use Raoultâs Law to relate y to x for each component in terms of its vapor pressure and the to-be-determined total pressure P.
Substitute the expressions you found for y into the material balances, which should each now have two unknowns --- x and P.
Solve for each x
Add the four expressions for the mole fractions x in the liquid.
Find P that makes the sum of the mole fractions in the liquid equal to 1.
Check your answer by confirming that sum of the mole fractions in the vapor also equal 1.