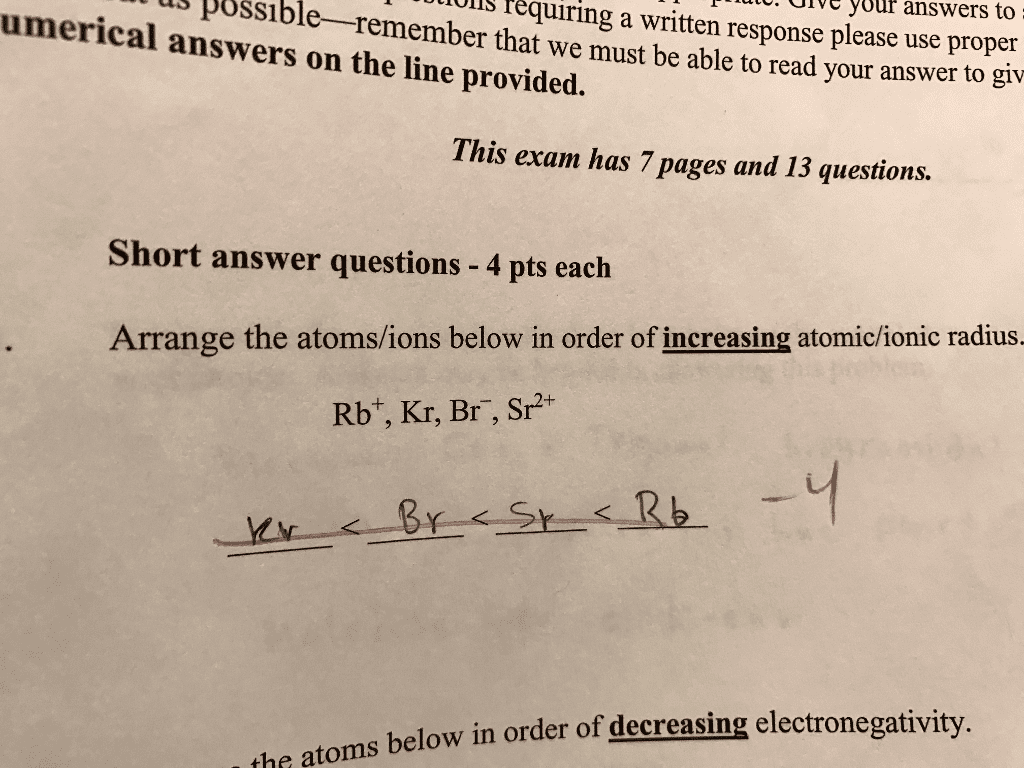1
answer
0
watching
312
views
6 Oct 2020
Arrange the elements Na, Si, and S in the order of
(a) decreasing atomic radius.
(b) increasing first ionization energy.
(c) decreasing electronegativity.
Arrange the elements Na, Si, and S in the order of
(a) decreasing atomic radius.
(b) increasing first ionization energy.
(c) decreasing electronegativity.
Rei Juez del MundoLv10
26 Nov 2020