1
answer
1
watching
439
views
6 Oct 2020
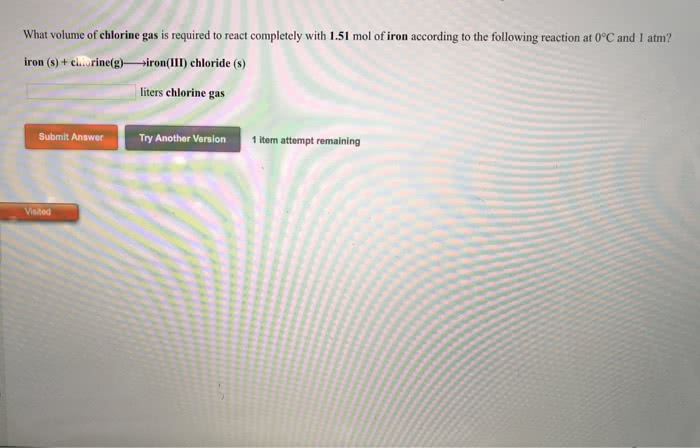
Iron reacts with chlorine gas to form iron (III) chloride. Given this, answer the following.
(a) What is the balanced chemical reaction?
(b) Given 10.0 g of iron, what mass, in grams, of chlorine gas would be required to complete the reaction?
(c) If only 18.5 g of FeCl3 is formed from 10.0 g of iron and excess chlorine gas, what is the percent yield of the reaction?
(d) If 10.0 g each of iron and chlorine gas are combined, what is the theoretical yield of iron (III) chloride?
Iron reacts with chlorine gas to form iron (III) chloride. Given this, answer the following.
(a) What is the balanced chemical reaction?
(b) Given 10.0 g of iron, what mass, in grams, of chlorine gas would be required to complete the reaction?
(c) If only 18.5 g of FeCl3 is formed from 10.0 g of iron and excess chlorine gas, what is the percent yield of the reaction?
(d) If 10.0 g each of iron and chlorine gas are combined, what is the theoretical yield of iron (III) chloride?
Brian HingpitLv10
5 Nov 2020