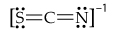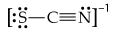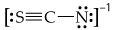1
answer
0
watching
519
views
6 Oct 2020
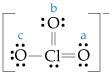
Consider the three possible structures of the thiocyanate ion (SCN-).
a. Draw the three resonance structures.
b. Calculate the formal charge on each atom in each resonance structure.
c. Based on formal charges and electronegativity, predict which resonance structure most closely approximates the bonding in this ion?
d. What are the similarities and differences of bonding in SCN- to the bonding in OCN-?
Consider the three possible structures of the thiocyanate ion (SCN-).
a. Draw the three resonance structures.
b. Calculate the formal charge on each atom in each resonance structure.
c. Based on formal charges and electronegativity, predict which resonance structure most closely approximates the bonding in this ion?
d. What are the similarities and differences of bonding in SCN- to the bonding in OCN-?
Greg MontoyaLv10
29 Oct 2020