2
answers
0
watching
334
views
6 Oct 2020
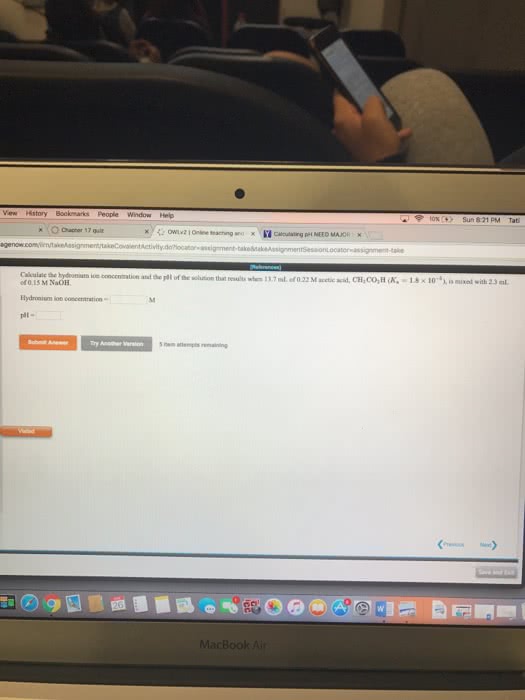
Calculate the hydronium ion concentration and pH of the solution that results when 22.0 mL of 0.15 M acetic acid (CH3CO2H) is mixed with 22.0 mL of 0.15 M NaOH.
Calculate the hydronium ion concentration and pH of the solution that results when 22.0 mL of 0.15 M acetic acid (CH3CO2H) is mixed with 22.0 mL of 0.15 M NaOH.
Read by 4 people
Neal BroomheadLv7
3 Nov 2020
Already have an account? Log in