1
answer
0
watching
273
views
26 Jul 2020
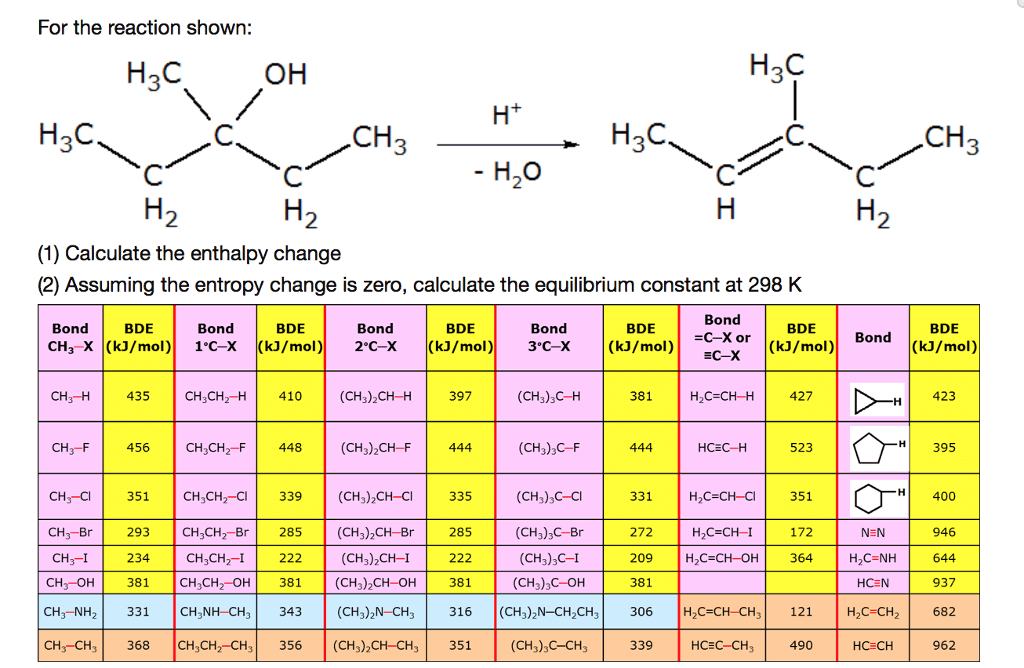
What is the enthalpy of the reaction shown below?

Bond
Bond Enthalpy, 
C-H
413
C-C
346
C=C
602
C≡C
835
C-Br
285
Br-Br
193
What is the enthalpy of the reaction shown below?
| Bond | Bond Enthalpy, |
| C-H | 413 |
| C-C | 346 |
| C=C | 602 |
| C≡C | 835 |
| C-Br | 285 |
| Br-Br | 193 |
1
answer
0
watching
273
views
For unlimited access to Homework Help, a Homework+ subscription is required.
1 Sep 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Using bondenergies, estimate the enthalpy change, in kJ, for the followinggas-phase reaction.
CH4(g)+ 2O2(g) ? CO2(g) +2H2O(g)
Bond Energies (inkJ/mol)
H-H | C-H | C-O | O-H | C-C | N-H | C-N | N-N | N-O | O-O | O-F | H-F |
432 | 411 | 358 | 459 | 346 | 386 | 305 | 167 | 201 | 142 | 190. | 565 |
C=C | C?C | C=O in CO2 | C?O | N=N | N?N | O=O |
602 | 835 | 799 | 1.072 x 1003 | 418 | 942 | 494 |
| -1.51 x 1003kJ |
| -588 kJ |
| 116 kJ |
| -802 kJ |
| -353 kJ |