1
answer
0
watching
181
views
25 Jul 2020
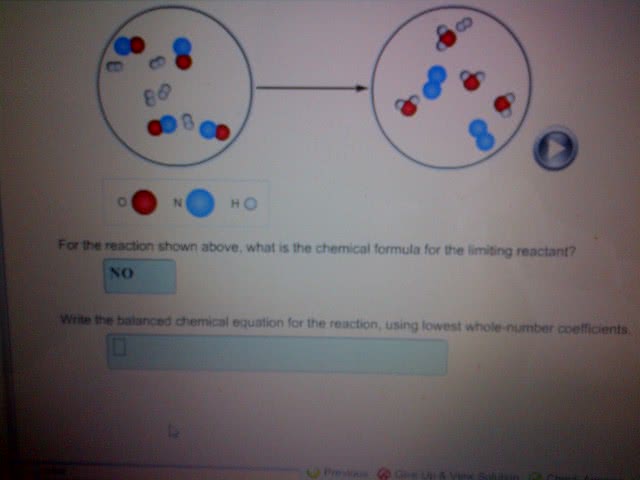
Calculate for the  given the following values and balanced chemical reaction.
given the following values and balanced chemical reaction.


Calculate for the given the following values and balanced chemical reaction.
1
answer
0
watching
181
views
For unlimited access to Homework Help, a Homework+ subscription is required.
1 Sep 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Why must chemical equations be balanced?
Choose all that apply.
Choose all that apply.
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict whether the given reaction is possible or not. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict whether the reaction requires a catalyst or not. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict the amount of product that can form from a given amount of reactant. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict how much of one reactant is required to react with a given amount of another. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict how much reactants are required to form a given amount of products. |