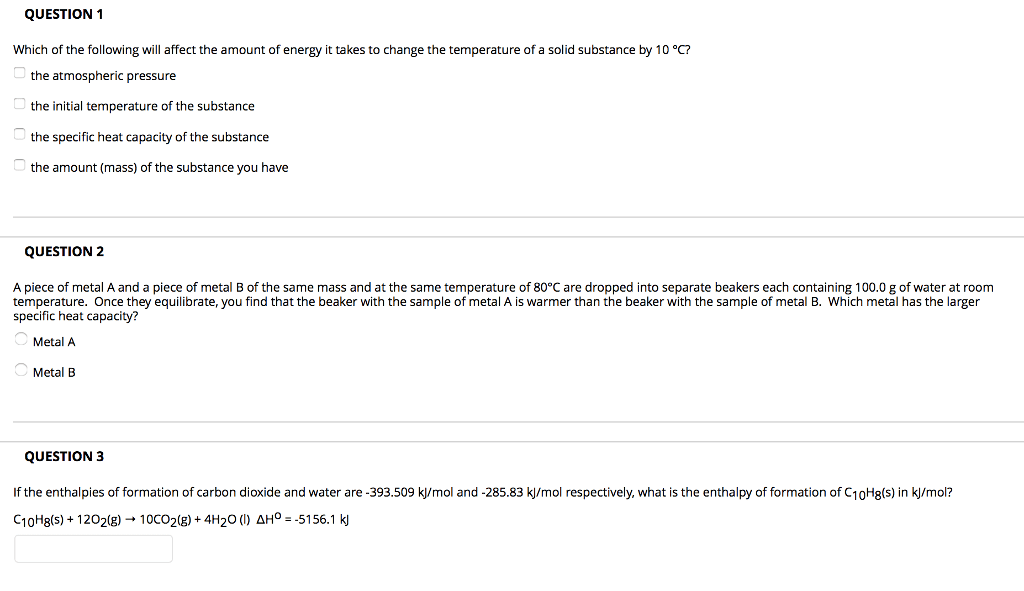
1
answer
0
watching
158
views
25 Jul 2020
Two metals of equal weight, X and Z, are heated to the  . Then, they were submerged into two separate beakers of negligible heat capacity with
. Then, they were submerged into two separate beakers of negligible heat capacity with  of
of  water. The temperature of the water in beaker X increased by
water. The temperature of the water in beaker X increased by  while the temperature of the water in beaker Z increased by
while the temperature of the water in beaker Z increased by  . With those in mind, which metal has a higher heat capacity? The specific heat of water is
. With those in mind, which metal has a higher heat capacity? The specific heat of water is  and the density is
and the density is  .
.
Two metals of equal weight, X and Z, are heated to the . Then, they were submerged into two separate beakers of negligible heat capacity with
of
water. The temperature of the water in beaker X increased by
while the temperature of the water in beaker Z increased by
. With those in mind, which metal has a higher heat capacity? The specific heat of water is
and the density is
.
1 Sep 2020