1
answer
0
watching
500
views
26 Jun 2020
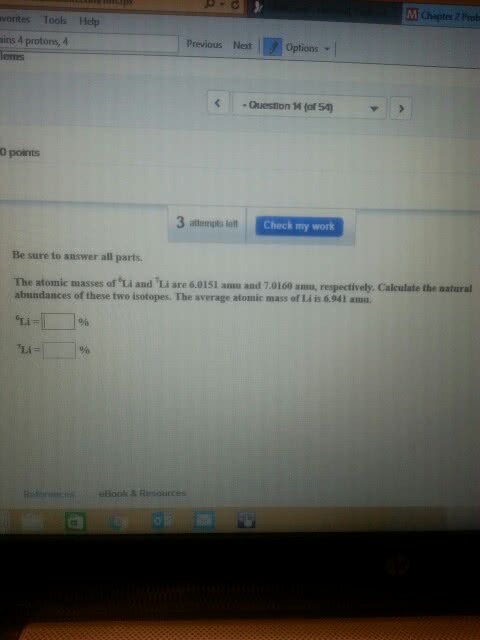
The element occurs in nature as two isotopes having atomic masses 10 u and 11 u. What are the percentage abundances of these isotopes in a sample of boron having an average atomic mass of 10.8 u?
The element occurs in nature as two isotopes having atomic masses 10 u and 11 u. What are the percentage abundances of these isotopes in a sample of boron having an average atomic mass of 10.8 u?
1 Sep 2020