1
answer
0
watching
78
views
25 Mar 2020
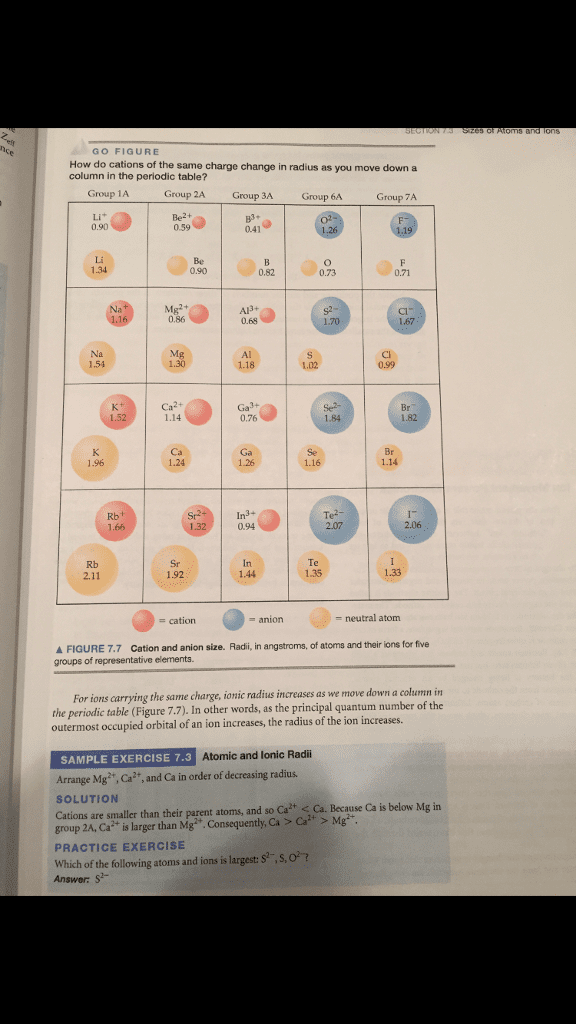
The atomic and ionic radii of the first three group 6A elements are

(a) Explain why the atomic radius increases in moving downward in the group. (b) Explain why the ionic radii are larger than the atomic radii. (c) Which of the three anions would you expect to be the strongest base in water? Explain.
The atomic and ionic radii of the first three group 6A elements are
(a) Explain why the atomic radius increases in moving downward in the group. (b) Explain why the ionic radii are larger than the atomic radii. (c) Which of the three anions would you expect to be the strongest base in water? Explain.
Irving HeathcoteLv2
26 May 2020