1
answer
0
watching
355
views
27 Apr 2020
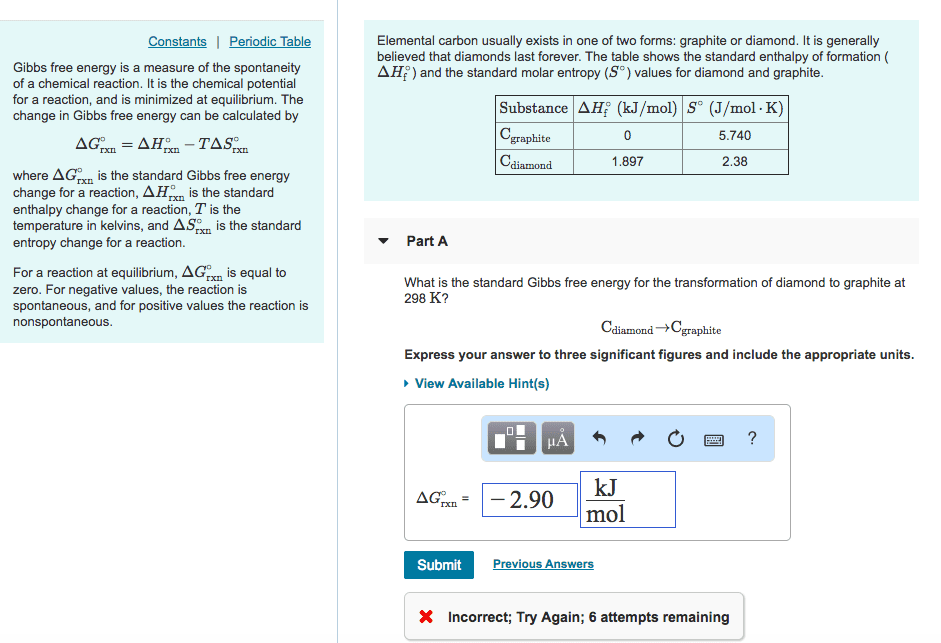
Three of the forms of elemental carbon are graphite, diamond, and buckminsterfullerene. The entropies at 298 K for graphite and diamond are listed in Appendix C. (a) Account for the difference in the S° values of graphite and diamond in light of their structures (Figure 12.29, p. 503). (b) What would you expect for the S° value of buckminsterfullerene (Figure 12.47, p. 516) relative to the values for graphite and diamond? Explain.
Three of the forms of elemental carbon are graphite, diamond, and buckminsterfullerene. The entropies at 298 K for graphite and diamond are listed in Appendix C. (a) Account for the difference in the S° values of graphite and diamond in light of their structures (Figure 12.29, p. 503). (b) What would you expect for the S° value of buckminsterfullerene (Figure 12.47, p. 516) relative to the values for graphite and diamond? Explain.
Patrina SchowalterLv2
29 May 2020