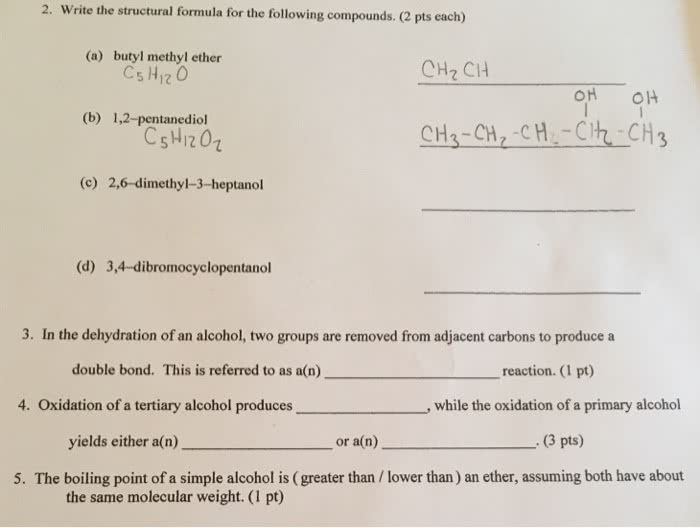2
answers
2
watching
1,225
views
ambermole700Lv1
30 Apr 2020
Propyl alcohol (CH3CH2CH2OH) and isopropyl alcohol  (CH3)2CHOH], whose space-filling models are shown, have boiling points of 97.2 and 82.5 °C, respectively. Explain why the boiling point of propyl alcohol is higher, even though both have the molecular formula, C3H8O.
(CH3)2CHOH], whose space-filling models are shown, have boiling points of 97.2 and 82.5 °C, respectively. Explain why the boiling point of propyl alcohol is higher, even though both have the molecular formula, C3H8O.

Propyl alcohol (CH3CH2CH2OH) and isopropyl alcohol (CH3)2CHOH], whose space-filling models are shown, have boiling points of 97.2 and 82.5 °C, respectively. Explain why the boiling point of propyl alcohol is higher, even though both have the molecular formula, C3H8O.
2
answers
2
watching
1,225
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Liked by diagupta003
Tod ThielLv2
13 May 2020
Read by 9 people
2 Jul 2021
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232