1
answer
0
watching
1,125
views
30 Mar 2020
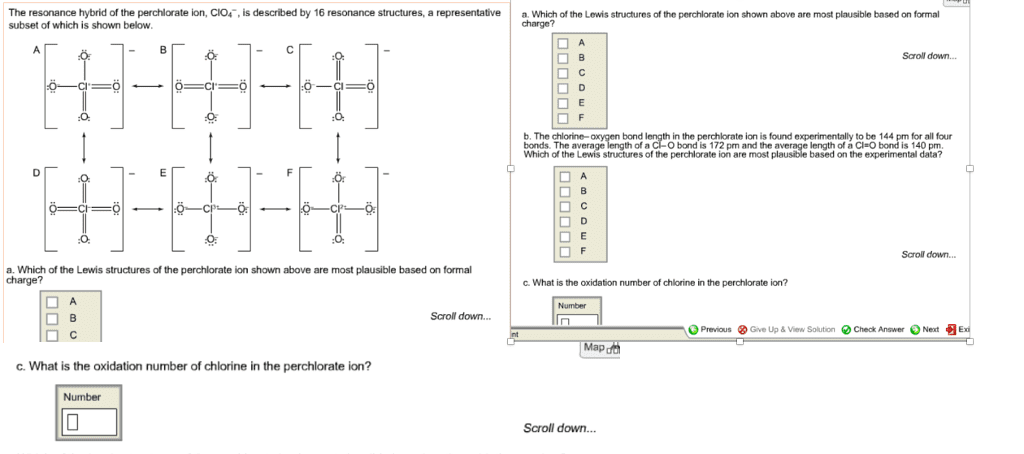
(a) Determine the formal charge on the chlorine atom in the hypochlorite ion, ClO-, and the perchlorate ion, ClO4-, using resonance structures where the Cl atom has an octet. (b) What are the oxidation numbers of chlorine in ClO- and in ClO4-? (c) Perchlorate is a much stronger oxidizing agent than hypochlorite. Suggest an explanation.
(a) Determine the formal charge on the chlorine atom in the hypochlorite ion, ClO-, and the perchlorate ion, ClO4-, using resonance structures where the Cl atom has an octet. (b) What are the oxidation numbers of chlorine in ClO- and in ClO4-? (c) Perchlorate is a much stronger oxidizing agent than hypochlorite. Suggest an explanation.
Hubert KochLv2
26 May 2020