1
answer
0
watching
243
views
20 Mar 2020
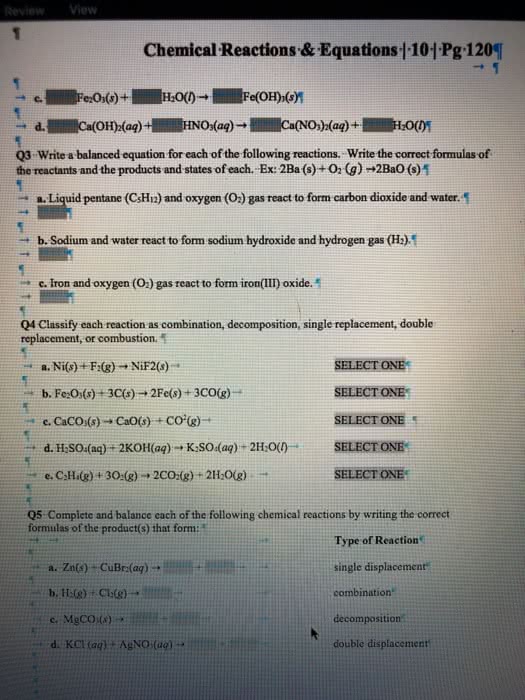
A key step in balancing chemical equations is correctly identifying the formulas of the reactants and products. For example, consider the reaction between calcium oxide, CaO(s), and H2O(l) to form aqueous calcium hydroxide. (a) Write a balanced chemical equation for this combination reaction, having correctly identified the product as Ca(OH)2(aq). (b) Is it possible to balance the equation if you incorrectly identify the product as CaOH(aq), and if so, what is the equation?
A key step in balancing chemical equations is correctly identifying the formulas of the reactants and products. For example, consider the reaction between calcium oxide, CaO(s), and H2O(l) to form aqueous calcium hydroxide. (a) Write a balanced chemical equation for this combination reaction, having correctly identified the product as Ca(OH)2(aq). (b) Is it possible to balance the equation if you incorrectly identify the product as CaOH(aq), and if so, what is the equation?
Tod ThielLv2
20 May 2020