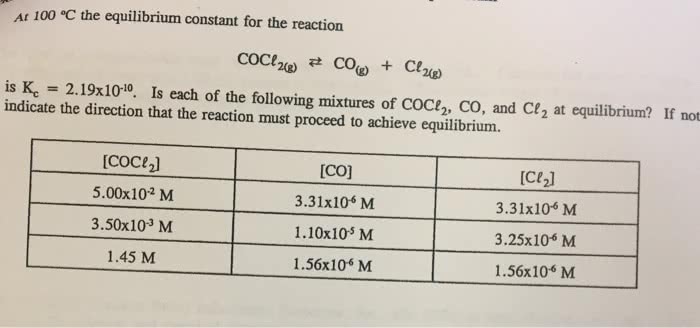1
answer
0
watching
232
views
whitebat914Lv1
21 Mar 2020
At 100 °C, the equilibrium constant for the reaction COCl2(g)  CO(g) + Cl2(g) has the value Kc = 2.19 × 10–10. Are the following mixtures of COCl2, CO, and Cl2 at
CO(g) + Cl2(g) has the value Kc = 2.19 × 10–10. Are the following mixtures of COCl2, CO, and Cl2 at
100 °C at equilibrium? If not, indicate the direction that the reaction must proceed to achieve equilibrium.
(a) [COCl2] = 2.00 × 10-3 M, [CO] = 3.3 × 10-6 M, [Cl2] = 6.62 × 10-6 M
(b) [COCl2] = 4.50 × 10-2 M, [CO] = 1.1 × 10-7 M, [Cl2] = 2.25 × 10-6 M
(c) [COCl2] = 0.0100 M, [CO] = [Cl2] = 1.48 × 10-6 M
At 100 °C, the equilibrium constant for the reaction COCl2(g) CO(g) + Cl2(g) has the value Kc = 2.19 × 10–10. Are the following mixtures of COCl2, CO, and Cl2 at
100 °C at equilibrium? If not, indicate the direction that the reaction must proceed to achieve equilibrium.
(a) [COCl2] = 2.00 × 10-3 M, [CO] = 3.3 × 10-6 M, [Cl2] = 6.62 × 10-6 M
(b) [COCl2] = 4.50 × 10-2 M, [CO] = 1.1 × 10-7 M, [Cl2] = 2.25 × 10-6 M
(c) [COCl2] = 0.0100 M, [CO] = [Cl2] = 1.48 × 10-6 M
Lelia LubowitzLv2
13 May 2020